
Concept explainers
(a)
Interpretation:
The product obtained by the reaction of the given
Concept Introduction:
Hydaration reaction is nothing but the addition of water to an alkene. In this reaction two bonds are simultaneously broken, one is carbon-carbon double bond, and second one is
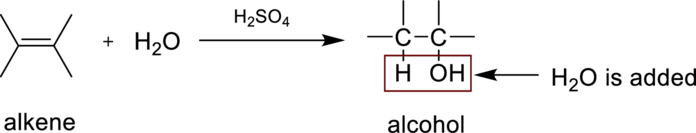
In the addition of water to an unsymmetrical alknene, the hydrogen from water is bonded to the less substituted carbon atom and is called Markovnikov’s rule.
(b)
Interpretation:
The product obtained by the reaction of the given alkene with water in the presence of sulfuric acid has to be identified.
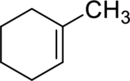
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
The product obtained by the reaction of the given alkene with water in the presence of sulfuric acid has to be identified.
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
Connect 1-Semester Online Access for Principles of General, Organic & Biochemistry
- Question 5: Draw the products when each alkene is treated with O3 followed by CH3SCH3. a. b. rad si snodls dos nosoarrow_forward1. Which alkene is the major product of this dehydration? OH H2SO4 heat A B Carrow_forwardConvert propan-2-ol [(CH3)2CHOH] to each compound. You may use any other organic or inorganic compounds.arrow_forward
- What alkenes are formed when each alcohol is treated with H 2SO 4? Use the Zaitsev rule to predict the major product.arrow_forwardPLEASE HELP Draw the products formed when cyclohexene is treated with each reagent.arrow_forwardWhat product is formed when each alkene is treated with H2 and a Pd catalyst? а. СНЗСН2CH-CHCH2CHЗ CH3 CH,CH(CH3)2 b. CH H. CH3 с.arrow_forward
- What alcohol is formed when each compound is treated with H 2 and a Pd catalyst?arrow_forwardGive the IUPAC name for each compound. a. H-C=C-CHC(CH,CH;CH3)3 b. CH3C= CC(CH3)CICH,CH3 c. CH2= CHCH;CH(CH,CH;)C = CC(CH)CH2CH;CH3arrow_forwardName each alkene: a. CH3CH=CHCH2CH2CH3 b. CH3CH=CH2 c. CH2CH3 l CH2=CHCH2CHCH3arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





