
Concept explainers
(a)
Interpretation:
Whether the given pair of compounds related as constitutional isomers, stereoisomers, or identical has to be indicated.
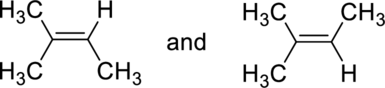
Concept Introduction:
Constitutional isomer is nothing but the structural isomer which has same molecular formula but differ in their connectivity of atoms.
Stereoisomers are spatial isomers which have same molecular formula and have differ in special arrangements which means the three-dimensional orientations of their atoms in space are arranged in different manner.
(b)
Interpretation:
Whether the given pair of compounds related as constitutional isomers, stereoisomers, or identical has to be indicated.
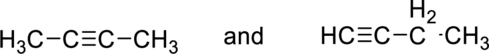
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
Connect 1-Semester Online Access for Principles of General, Organic & Biochemistry
- 2-methylhexane & 3,3-dimethylpentane Are the two compounds Constitutional Isomers or Different Compounds? Draw structures for both compounds.arrow_forwardExplain the concept of Cyclohexane Conformations ?arrow_forwardClassify each pair of compounds as constitutional isomers or identical molecules.arrow_forward
- Five structural isomers, or constitutional isomers, have the formula C6H14C6H14. Draw the indicated isomers, grouped by number of carbon atoms in the main chain. a) Draw two isomers with 5 carbon atoms in the main chain. Be sure to include all hydrogen atoms. b) Draw two isomers with 4 carbon atoms in the main chain. Be sure to include all hydrogen atoms.arrow_forwardDistinguish between isomerism and resonance. Distinguish between structural and geometric isomerism. When writing the various structural isomers, the most difficult task is identifying which are different isomers and which are identical to a previously written structurethat is, which are compounds that differ only by the rotation of a carbon single bond. How do you distinguish between structural isomers and those that are identical? Alkenes and cycloalkanes are structural isomers of each other. Give an example of each using C4H8. Another common feature of alkenes and cycloalkanes is that both have restricted rotation about one or more bonds in the compound, so both can exhibit cis- trans isomerism. What is required for an alkene or cycloalkane to exhibit cis-trans isomerism? Explain the difference between cis and trans isomers. Alcohols and ethers are structural isomers of each other, as are aldehydes and ketones. Give an example of each to illustrate. Which functional group in Table 21-4 can be structural isomers of carboxylic acids? What is optical isomerism? What do you look for to determine whether an organic compound exhibits optical isomerism? 1-Bromo-1-chloroethane is optically active whereas 1-bromo-2-chloroethane is not optically active. Explain.arrow_forwardWhy are different conformations of an alkane not considered structural isomers?arrow_forward
- a) Draw and name the eight isomeric alcohols with the formula C5H12O b) Classify each of the alcohols in a) as 1º, 2º, or 3⁰.arrow_forwardDraw the structure of all compounds that fit the following descriptions. a. Five constitutional isomers having the molecular formula C4Hg.arrow_forwardThe octane rating for gasoline is a measurement of how readily a fuelcombusts compared to 2,2,4-trimethylpentane, an isomer of octane.a. Draw 2,2,4-trimethylpentane and verify that it is an isomer ofoctane.b. Draw four other isomers of octane.c. Select one of the isomers and draw it such that it looksdifferent on the page but is still the exact same compound.d. Name this isomer.e. Define isomer using a complete sentence.arrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





