
Concept explainers
(a)
Interpretation:
The product formed from the condensation reaction of butanoic acid and 2-propanol has to be drawn.
Concept Introduction:
Ester:
The structure of an ester is,
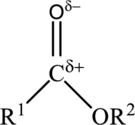
The carbonyl group of the ester is polar and could participate in dipole-dipole attractions. Hydrocarbon chains flank the carbonyl group
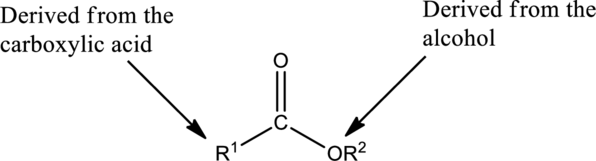
Preparation of Esters:
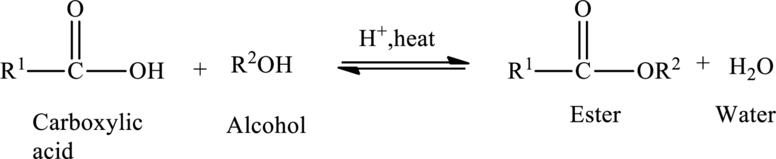
Esters are prepared from the reaction of
The general preparation of esters is shown below,
(b)
Interpretation:
The product formed from the condensation reaction of ethanoic acid and 1-pentanol has to be drawn.
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
The product formed from the condensation reaction of hexanoic acid and methylethyl
Concept Introduction:
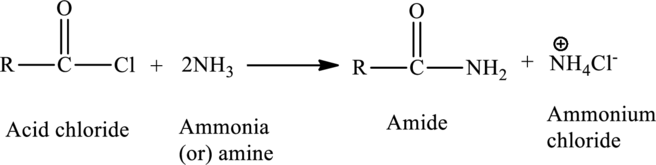
Amides can be prepared in a reaction between a carboxylic acid and an amine. The acid derivative can be either an acid chloride or an acid anhydride.
The general reaction is,
(d)
Interpretation:
The product formed from the condensation reaction of ethanoic acid and propylamine has to be drawn.
Concept Introduction:
Refer to part (d).
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
ACHIEVE/CHEMICAL PRINCIPLES ACCESS 1TERM
- Give the structural formulae and name the functional groups of the following compounds. (a) 3-chlorobut-1-ene Name the functional group: (b) butanedioic acid Name the functional group: (c) propanamide Name the functional group: (d) 3-methylbutanal Name the functional group:arrow_forwardDraw the product resulting from mild oxidation of (a) 2-butanol; (b) 2-methylpropanal; (c) cyclopentanol.arrow_forwardDraw the organic product formed when the followingcompounds undergo a substitution reaction: (a) acetic acid and1-hexanol; (b) propanoic acid and dimethylamine; (c) ethanoicacid and diethylamine.arrow_forward
- The reactant: benzophenone & 2-propanol the product: benzopinacol & acetonearrow_forwardWrite the equations for the preparation of alcohols by reacting a) propene with sulfuric acid and water b) chlorocyclohexane and sodium hydroxide Write the equations for the reaction of phenoxybenzene with hot concentrated HCIarrow_forward5. Give the structural formulae and name the functional groups of the following compounds. (a) 3-chlorobut-1-ene (b) butanedioic acid Name the functional group: (c) propanamide Name the functional group: (d) 3-methylbutanal Name the functional group: Name the functional group:arrow_forward
- Draw the structures of the following compounds:(a) Ethanoic acid(b) Bromopentane(c) Butanonearrow_forwardIdentify which of the following statement(s) is/are true. (f) Aldehydes, ketones, carboxylic acids, and esters all contain a carbonyl group. (g) A compound with the molecular formula of C3H6O may be either an aldehyde, a ketone, or a carboxylic acid. (h) Bond angles about the carbonyl carbon of an aldehyde, a ketone, a carboxylic acid, and an ester are all approximately 109.5°. (i) The molecular formula of the smallest aldehyde is C3H6O, and that of the smallest ketone is also C3H6O. (j) The molecular formula of the smallest carboxylic acid is C2H4O2.arrow_forward(a) Write a chemical test to distinguish between: (i) Chlorobenzene and Benzyl chloride. (ii) Chloroform and Carbon tetrachloride. (b) Why is methyl chloride hydrolysed more easily than chlorobenzene?arrow_forward
- 5.Write the structural formula of the ester that, when hydrolyzed, would yield the following:(a) methanol and propanoic acid(b) 1-octanol and acetic acid (c) ethanol and butanoic acidarrow_forwardWhat is the structure of the principal organic product formed in the reaction of 1-iodopropane with KOCH(CH3)2?arrow_forwardA cyclic, non-aromatic compound containing an attached carboxyl group is named by naming the cyclic compound exclusive of the –COOH group and then adding the suffix - carboxylic acid. Draw the structure of cis-2-methylcyclopentanecarboxylic acid.arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

