
ORGANIC CHEMISTRY (LL) W/WILEYPLUS NEXT
12th Edition
ISBN: 9781119664635
Author: Solomons
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 11, Problem 11PP
Practice Problem 11.11
An exception to what is stated above has to do with syntheses of unsymmetrical ethers in which one alkyl group is a tert-butyl group and the other group is primary. For example, this synthesis can be accomplished by adding tm-butyl alcohol to a mixture of the primary alcohol and
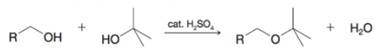
Give a likely mechanism for this reaction and explain why it is successful.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Using your reaction roadmap as a guide, show how to convert 1-butanol and ethanol into racemic 2-ethoxy-1-butanol. You must use 1-butanol and ethanol as the source of
all carbon atoms in the ether product.
(1)
(2)
(3)
OH
(4)
2-ethoxy-1-butanol
(racemic)
OH
Suggest reagents and experimental conditions for each step in this synthesis.
From the choices provided, suggest appropriate reagents for each step. More than one reagent may be necessary. Use the minimum number of steps possible. Enter your
answer as a letter, or a series of letters, in the order necessary to bring about the steps shown.
Reagents:
OH
(c) ethylene oxide
(a) (CH3)3CO¯K+, THF (d) H₂SO4, H₂O
(b) OsO4, H₂O2
Br
OH
+
(9) H₂ SO4 (anhydrous) Na, NH3 (1)
(k) NBS, heat
(e) m-chloroperoxybenzoic acid
(mCPBA)
(f) pyridinium chlorochromate) PBr3
(PCC)
(h) NaOH
(N-bromosuccinimide)
Previous
Next
Using your reaction roadmap as a guide, show how to convert 1-butanol and ethanol into racemic 2-ethoxy-1-butanol. You must use 1-butanol and ethanol as the source of
all carbon atoms in the ether product.
(1)
(2)
(3)
OH
(4)
2-ethoxy-1-butanol
(racemic)
OH
OH
Suggest reagents and experimental conditions for each step in this synthesis.
From the choices provided, suggest appropriate reagents for each step. More than one reagent may be necessary. Use the minimum number of steps possible. Enter your
answer as a letter, or a series of letters, in the order necessary to bring about the steps shown.
Reagents:
1
(a) (CH3)3 CO K+, THF (d) H₂SO4, H₂O
(b) OsO4, H₂O2
(c) ethylene oxide
Br
(e) m-chloroperoxybenzoic acid
OH
(mCPBA)
(f) pyridinium chlorochromate
(PCC)
|| Ⓡ
(g) H₂SO4 (anhydrous)
(h) NaOH
(i) PBr3
(j) Na, NH3 (1)
(k) NBS, heat
(N-bromosuccinimide)
Previous
Next
Using your reaction roadmap as a guide, show how to convert 1-butanol and ethanol into racemic 2-ethoxy-1-butanol. You must use 1-butanol and ethanol as the source of
all carbon atoms in the ether product.
(1)
(2)
(3)
OH
(4)
2-ethoxy-1-butanol
(racemic)
OH
OII..
Suggest reagents and experimental conditions for each step in this synthesis.
From the choices provided, suggest appropriate reagents for each step. More than one reagent may be necessary. Use the minimum number of steps possible. Enter your
answer as a letter, or a series of letters, in the order necessary to bring about the steps shown.
Reagents:
(c) ethylene oxide
OH
(a) (CH3)3 CO K+, THF (d) H₂SO4, H₂O
(b) OsO4, H₂O2
Br
OH
+
(9) H₂ SO4 (anhydrous) Na, NH3 (1)
(k) NBS, heat
(e) m-chloroperoxybenzoic acid
(mCPBA)
(f) pyridinium chlorochromatę) PBr3
(PCC)
(h) NaOH
(N-bromosuccinimide)
Previous
Next
Chapter 11 Solutions
ORGANIC CHEMISTRY (LL) W/WILEYPLUS NEXT
Ch. 11 - Prob. 1PPCh. 11 - Practice Problem 11.2 Give bond-line formulas and...Ch. 11 - Prob. 3PPCh. 11 - Prob. 4PPCh. 11 - Prob. 5PPCh. 11 - Prob. 6PPCh. 11 - Prob. 7PPCh. 11 - Practice Problem 11.8 Show how you would prepare...Ch. 11 - Prob. 9PPCh. 11 - Prob. 10PP
Ch. 11 - Practice Problem 11.11
An exception to what is...Ch. 11 - Prob. 12PPCh. 11 - Prob. 13PPCh. 11 - Prob. 14PPCh. 11 - Prob. 15PPCh. 11 - Prob. 16PPCh. 11 - Prob. 17PPCh. 11 - Prob. 18PPCh. 11 - Practice Problem 11.19
Propose structures for each...Ch. 11 - Prob. 20PPCh. 11 - Prob. 21PPCh. 11 - Prob. 22PPCh. 11 - Prob. 23PPCh. 11 - Prob. 24PPCh. 11 - Give an IUPAC substitutive name for each of the...Ch. 11 - Prob. 26PCh. 11 - Prob. 27PCh. 11 - Prob. 28PCh. 11 - Prob. 29PCh. 11 - 11.30. Show how you might prepare 2-bromobutane...Ch. 11 - Prob. 31PCh. 11 - Prob. 32PCh. 11 - Prob. 33PCh. 11 - Considering A-L to represent the major products...Ch. 11 - Write structures for the products that would be...Ch. 11 - Prob. 36PCh. 11 - Provide the reagents necessary for the following...Ch. 11 - 11.38. Predict the major product from each of the...Ch. 11 - Predict the products from each of the following...Ch. 11 - Provide the reagents necessary to accomplish the...Ch. 11 - 11.41. Provide reagents that would accomplish the...Ch. 11 - Prob. 42PCh. 11 - 11.43. A synthesis of the -receptor blocker called...Ch. 11 - Prob. 44PCh. 11 - Prob. 45PCh. 11 - 11.46. For each of the following, write a...Ch. 11 - 11.47. Vicinal halo alcohols (halohydrins) can be...Ch. 11 - Prob. 48PCh. 11 - Prob. 49PCh. 11 - Prob. 50PCh. 11 - Prob. 51PCh. 11 - Prob. 52PCh. 11 - Outlined below is a synthesis of the gypsy moth...Ch. 11 - Prob. 54PCh. 11 - Prob. 55PCh. 11 - Prob. 56PCh. 11 - 11.57. When the 3-bromo-2-butanol with the...Ch. 11 - 11.58. Reaction of an alcohol with thionyl...Ch. 11 - Prob. 59PCh. 11 - Prob. 60PCh. 11 - Prob. 1LGPCh. 11 - Prob. 2LGPCh. 11 - Synthesize the compound shown below from...
Additional Science Textbook Solutions
Find more solutions based on key concepts
PRACTICE 1.3 The melting point of table salt is 1474oF. What temperature is this on the Celsius and Kelvin scal...
CHEMISTRY-TEXT
Draw the following orbitals: a. 3s orbital b. 4s orbital c. 3p orbital
Organic Chemistry (8th Edition)
Method to determine the charge on a transition metal cation from chemical formula is to be explained. Concept i...
Living By Chemistry: First Edition Textbook
16.43 The following pictures represent solutions at various stages in thetitration of a weak diprotic acid with...
Chemistry (7th Edition)
For each of the following, (i) give the systematic name of the compound and specify the oxidation state of the ...
General Chemistry: Atoms First
50. A 5.00-mL sample of an solution of unknown concentration is titrated with a 0.1090 M NaOH solution. A volu...
Introductory Chemistry (5th Edition) (Standalone Book)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Using your reaction roadmap as a guide, show how to convert 1-butanol and ethanol into racemic 2-ethoxy-1-butanol. You must use 1-butanol and ethanol as the source of all carbon atoms in the ether product. (1) (2) (3) OH (4) 2-ethoxy-1-butanol (racemic) OH OH (c) ethylene oxide Suggest reagents and experimental conditions for each step in this synthesis. From the choices provided, suggest appropriate reagents for each step. More than one reagent may be necessary. Use the minimum number of steps possible. Enter your answer as a letter, or a series of letters, in the order necessary to bring about the steps shown. Reagents: (a) (CH3)3 CO K+, THF (d) H₂SO4, H₂O (b) OsO4, H₂O2 1 Br (e) m-chloroperoxybenzoic acid OH (mCPBA) (f) pyridinium chlorochromate (PCC) + le 2 (9) H₂SO4 (anhydrous) (j) Na, NH3 (1) (h) NaOH (k) NBS, heat (i) PBr3 (N-bromosuccinimide) Previous Nextarrow_forwardUsing your reaction roadmap as a guide, show how to convert 1-butanol and ethanol into racemic 2-ethoxy-1-butanol. You must use 1-butanol and ethanol as the source of all carbon atoms in the ether product. (1) (2) (3) OH (4) 2-ethoxy-1-butanol (racemic) OH OH Suggest reagents and experimental conditions for each step in this synthesis. From the choices provided, suggest appropriate reagents for each step. More than one reagent may be necessary. Use the minimum number of steps possible. Enter your answer as a letter, or a series of letters, in the order necessary to bring about the steps shown. Reagents: (c) ethylene oxide (a) (CH3)3 COK, THF (d) H₂SO4, H₂O (b) OsO4, H₂O2 Br OH (e) m-chloroperoxybenzoic acid (mCPBA) (f) pyridinium chlorochromate (PCC) + 2 (9) H₂SO4 (anhydrous) (h) NaOH (i) PBr3 (j) Na, NH3 (1) (k) NBS, heat (N-bromosuccinimide) Previous Nextarrow_forwardUsing your reaction roadmap as a guide, show how to convert 1-butanol and ethanol into racemic 2-ethoxy-1-butanol. You must use 1-butanol and ethanol as the source of all carbon atoms in the ether product. (1) (2) (3) OH (4) 2-ethoxy-1-butanol (racemic) OH OH (c) ethylene oxide Suggest reagents and experimental conditions for each step in this synthesis. From the choices provided, suggest appropriate reagents for each step. More than one reagent may be necessary. Use the minimum number of steps possible. Enter your answer as a letter, or a series of letters, in the order necessary to bring about the steps shown. Reagents: (a) (CH3)3 COK+, THF (d) H₂SO4, H₂O (b) OsO4, H₂O2 Br OH (e) m-chloroperoxybenzoic acid (mCPBA) (f) pyridinium chlorochromate (PCC) + 2 Oly (i) PBr3 (9) H₂SO4 (anhydrous) (h) NaOH (j) Na, NH3 (1) (k) NBS, heat (N-bromosuccinimide) Previous Next Save and Exit ?arrow_forward
- we know that ethers, such as diethyl ether and tetrahydrofuran, are quite resistant to the action of dilute acids and require hot concentrated HI or HBr for cleavage. However, acetals in which two ether groups are linked to the same carbon undergo hydrolysis readily, even in dilute aqueous acid. How do you account for this marked difference in chemical reactivity toward dilute aqueous acid between ethers and acetals?arrow_forwardChemistry Provide reagents/conditions to accomplish the following syntheses. Several steps are required in some cases.arrow_forward7. The following reaction is from a synthesis of the natural product salinipyrone A. After being synthesized, salinipyrone A was then screened for antitumor activity. Predict the major product of this reaction. Note that the TBS-protected alcohol is stable to the Wittig reaction condition --1 الله OTBS Me OTBS - 1-0-4-1-84 Si-t-Bu Me H Ph₂P=CHCO₂Etarrow_forward
- 9. Plan syntheses of the following compounds. You may use the given starting material and any compound containing three or fewer carbons. (a) (b) (c) (d) by Br or Br H Br H OH = OHarrow_forwardThe reaction of 2,2-dimethyl-1-propanol [(CH3)3CCH2OH], also known by the common name neopentyl alcohol, with HBr is very slow and gives 2-bromo-2-methylbutane as the major product.Give a mechanistic explanation for these observations.arrow_forwardUsing your reaction roadmap as a guide, show how to convert cyclohexanol into racemic trans-1,2-cyclohexanediol. (1) (2) OH aa OH (3) trans-1,2-cyclohexanediol (racemic) OH "ОН Suggest reagents and experimental conditions for each step in this synthesis. From the choices provided, suggest appropriate reagents for each step. More than one reagent may be necessary. Use the minimum number of steps possible. Enter your answer as a letter, or a series of letters, in the order necessary to bring about the steps shown. Reagents: (c) ethylene oxide 1 OH (a) CH3ONa+, CH, OH (d) H₂SO4, H₂O (b) OsO4, H₂O2 ||Ⓡ (9) H₂SO4 (anhydrou$)) Na, NH3 (1) (k) NBS, heat (e) m-chloroperoxybenzoic acid (mCPBA) (f) pyridinium chlorochromatę) PBr3 (PCC) (h) NaOH (N-bromosuccinimide)arrow_forward
- Br2, H3O+ གོ་ཤི་ན་ཤི་ H3C CH3 CH3 CH3 Aldehydes and ketones can be halogenated at their a-position by reaction with Cl₂, Br2, or 12, under acidic conditions. Using Br2 under acidic conditions, an intermediate enol is formed which adds bromine at the a-position. The reaction stops after the addition of one bromine because the electron-withdrawing halogen decreases the basicity of the carbonyl oxygen, making the protonation less favorable. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions :OH :OH Br-Br H3C H3C. Br Br CH₂ H₂ H3C CH3 H3C CH3 2barrow_forward2B Suggest a short synthetic route for the preparation of compound D from compound C OH Br COOH C D Note: Apart from compound C, you can also use organic reagents with up to 1 C atom. The number of arrows in the figure above does not necessarily correspond to the number of steps.arrow_forward(a) Tsomane and Nyiko were given a task of synthesising methylenecyclohexane 2. After a brief discussion with each other, Tsomane proposed Method A to synthesise 2 from cyclohexanone 1 while Nyiko proposed Method B that started from hydroxymethylcyclohexane 3. Each student believed that their proposed method is better than the other. (Scheme below) (1) Ph Ph 8*8 Ph THF A 1 Santande B H₂SO4 100 °C 3 OH Using curly arrows, provide full mechanistic details accounting how methylenecyclohexane 2 was synthesised according to both Methods A and B.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Alcohols, Ethers, and Epoxides: Crash Course Organic Chemistry #24; Author: Crash Course;https://www.youtube.com/watch?v=j04zMFwDeDU;License: Standard YouTube License, CC-BY