
Concept explainers
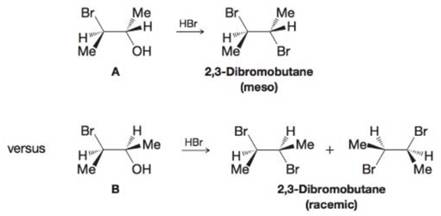
When the 3-bromo-2-butanol with the stereochemical structure A is treated with concentrated HBr, it yields meso-2, 3-dibromobutane; a similar reaction of the 3-bromo-2-butanol B yields
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
ORGANIC CHEMISTRY (LL) W/WILEYPLUS NEXT
Additional Science Textbook Solutions
Introductory Chemistry (6th Edition)
Chemistry: A Molecular Approach
Chemistry & Chemical Reactivity
Fundamentals of Heat and Mass Transfer
CHEMISTRY-TEXT
Chemistry: The Central Science (13th Edition)
- The base-promoted rearrangement of an -haloketone to a carboxylic acid, known as the Favorskii rearrangement, is illustrated by the conversion of 2-chlorocyclohexanone to cyclopentanecarboxylic acid. It is proposed that NaOH first converts the a-haloketone to the substituted cyclopropanone shown in brackets and then to the sodium salt of cyclopentanecarboxylic acid. (a) Propose a mechanism for base-promoted conversion of 2-chlorocyclohexanone to the proposed intermediate. (b) Propose a mechanism for base-promoted conversion of the proposed intermediate to sodium cyclopentanecarboxylate.arrow_forwardWhen warmed in dilute sulfuric acid, 1-phenyl-1,2-propanediol undergoes dehydration and rearrangement to give 2-phenylpropanal. (a) Propose a mechanism for this example of a pinacol rearrangement (Section 10.7). (b) Account for the fact that 2-phenylpropanal is formed rather than its constitutional isomer, 1-phenyl-1-propanone.arrow_forwardWhat would be the product if (S)-2-bromobutane underwent an S№1 reaction with water in the presence of sodium carbonate (a weak base)? (R)-2-butanol (S)-2-butanol A mixture of (R)- and (S)-2-butanolarrow_forward
- 7. Compound X is an alcohol. Upon oxidation by acidify potassium permanganate, a ketone, Y is formed. X reacts with phosphorous bromide (PBR3) to give Z which is then purified and reflux in dry ether along with Mg metal to form a Grignard reagent, S. S is then treated with Y to yield 3,4-di methyl-3-hexanol after hydrolysis in dilute HCI. Identify X,Y, Z and S. Write all the chemical equations for the reaction involved.arrow_forwardIn the Grignard Reaction experiment what would the reaction mechanism be if the reactants are Magnesium, 2-bromopropane, 4-benzaldehyde, phosphoric acid and diethyl ether, resulting in a product of 1-(4-methoxyphenyl)-2-methylpropan-1-olarrow_forwardWhat factor seems to be responsible for the Wagner-Meerwein rearrangement of camphene to give isobornyl acetate (a 3º → 2º carbocation rearrangement)?arrow_forward
- (A true story.) A drug user responded to an ad placed by a DEA informant in a drug-culture magazine. He later flew fromColorado to Maryland, where he bought some 1-phenyl-2-propanone (P2P) from the informant. The police waited nearlya month for the suspect to synthesize something, then obtained a search warrant, and searched the residence. They foundthe unopened bottle of P2P; apparently, the suspect was not a good chemist and was unable to follow the instructions theinformant gave him. They also found pipes and bongs with residues of marijuana and cocaine, plus a bottle of methylamine hydrochloride, some muriatic acid (dilute HCl), zinc strips, flasks, and other equipment.(a) Assume you are consulting for the police. Show what synthesis the suspect was prepared to carry out, to provideprobable cause for the charge of attempting to manufacture a controlled substance.(b) Assume you are a member of the jury. Would you convict the defendant of attempting to manufacture a…arrow_forwardWhat reaction would be best for preparing 2-isopropoxy pentane in high yield? 2-iodopentane and sodium isopropoxide sodium hydride added to 2-pentanol, then 2-iodopropane mercury acetate added to 1-pentene and isopropanol, then NaBH4 mercury acetate added to 2-pentene and isopropanol, then NaBH4arrow_forwardProvide the reagents for the short syntheses (about 3-4 steps).arrow_forward
- 7. Compound EE, C4H3O shows a positive result (formation of yellow precipitate) when reacted with lodoform reagent. Reduction of compound EE with sodium boron hydride, NABH4 followed by acidified water, H3O* produces an alcohol FF. Compound FF is treated with phosporus pentachloride, PCI5 to form compound GG. Prolong heating of compound EE with concentrated solution of acidified potassium permanganate, KMNO4 will form compound HH and II. Sebatian EE, CaHạO menunjukkan keputusan positif (pembentukkan mendakan kuning) apabila bertindak balas dengan reagen lodoform. Penurunan sebatian EE dengan natrium boron hidrida, NABH4 diikuti dengan air berasid, H3O* menghasilkan satu alkohol FF. Sebatian FF telah dirawat dengan fosforus pentaklorida, PCI5 untuk membentuk sebatian GG. Pemanasan yang berterusan bagi sebatian EE dengan larutan pekat kalium permanganat, KMNO4 berasid akan membentuk sebatian HH dan II. Draw the structural formula of EE, FF, GG, HH and II. Lukiskan formula struktur bagi…arrow_forwardShow with examples and structures the Order of Reactivity in SN2. 2- Show Some SN2 Reactions With Bromo-Ethane. Give 9 examples of Nucleophiles Nu-. 3- Rank the following compounds in order of their expected reactivity toward SN2 reaction: CH3Br, CH3OTos, (CH3)2CHCl 4- Show with examples for the Influence of Solvents in the SN2 Reaction. 5- Show the mechanism of the SN1 Reaction with the proper directions of the arrows.arrow_forward2-Chlorobutane reacts much more slowly than 2-chloro-2-methylpropane in the silver nitrate test. Explain this difference in reactivity.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
