
ORGANIC CHEMISTRY-W/STUD.SOLN.MAN.
10th Edition
ISBN: 9781260001099
Author: Carey
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 11, Problem 37P
Identify the more reactive dienophile in each of the following pairs.
a) 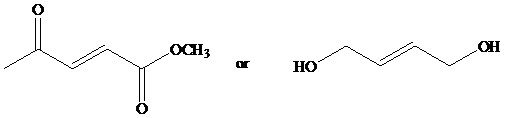
b) 
c) 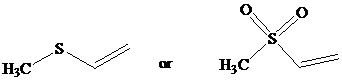
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
For the following reactions:a. Provide the missing major product, OR, missing reagent and conditions appropriate.b. Pay attention to stereochemistry unless directed otherwise and identify any racemicmixture using the symbol (±)c. Assign each reaction as addition, elimination, substitution or rearrangement, unlessdirected otherwise.
a) place asterisks(*) at the allylic position of compound A.
b) Draw the resonance forms of the allylic radical intermediate that accounts for the formation of B and C.
Identify the pericyclic reactions in the followingreaction schemes. Give the complete reaction name and indicate the course of the reaction with the aid of the arrow notation.
Chapter 11 Solutions
ORGANIC CHEMISTRY-W/STUD.SOLN.MAN.
Ch. 11.1 - Prob. 1PCh. 11.1 - Prob. 2PCh. 11.2 - The two compounds shown differ by a factor of 60...Ch. 11.2 - Prob. 4PCh. 11.2 - Prob. 5PCh. 11.2 - Prob. 6PCh. 11.3 - Prob. 7PCh. 11.3 - Prob. 8PCh. 11.3 - Evaluate 2,3,3-trimethyl-1-butene as a candidate...Ch. 11.4 - After heating a solution of allyl tert-butyl...
Ch. 11.5 - Prob. 11PCh. 11.6 - Prob. 12PCh. 11.8 - Prob. 13PCh. 11.9 - What dienes containing isolated double bonds are...Ch. 11.10 - Prob. 15PCh. 11.10 - Prob. 16PCh. 11.11 - Prob. 17PCh. 11.12 - Dicarbonyl compounds such as quinones are reactive...Ch. 11.12 - 2,3-Di-tert-butyl-1,3-butadiene is extremely...Ch. 11.12 - Methyl acrylate (H2C=CHCO2CH3) reacts with...Ch. 11.13 - Prob. 21PCh. 11.14 - What diene and dienophile could you use to prepare...Ch. 11.14 - Write equations in the synthetic direction for the...Ch. 11.16 - Prob. 24PCh. 11.16 - Prob. 25PCh. 11 - Write structural formulas for each of the...Ch. 11 - Give an acceptable IUPAC name for each of the...Ch. 11 - A certain species of grasshopper secretes an...Ch. 11 - Which of the following are chiral?...Ch. 11 - Describe the molecular geometry expected for...Ch. 11 - Prob. 31PCh. 11 - What compound of molecular formula C6H10 gives...Ch. 11 - Prob. 33PCh. 11 - Prob. 34PCh. 11 - Prob. 35PCh. 11 - Prob. 36PCh. 11 - Identify the more reactive dienophile in each of...Ch. 11 - Prob. 38PCh. 11 - Prob. 39PCh. 11 - Prob. 40PCh. 11 - Prob. 41PCh. 11 - Prob. 42PCh. 11 - Prob. 43PCh. 11 - Prob. 44PCh. 11 - Prob. 45PCh. 11 - Prob. 46PCh. 11 - Show how to prepare each of the following...Ch. 11 - Prob. 48PCh. 11 - Prob. 49PCh. 11 - Prob. 50PCh. 11 - Compound A was converted to compound B by the...Ch. 11 - Suggest reasonable explanations for each of the...Ch. 11 - Prob. 53PCh. 11 - Prob. 54PCh. 11 - Prob. 55DSPCh. 11 - Prob. 56DSPCh. 11 - Prob. 57DSPCh. 11 - Prob. 58DSPCh. 11 - Prob. 59DSP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Provide the missing starting material, reactant or product. Show appropriate stereochemistry. a) b)arrow_forward(A) Provide the major organic product for the reaction below (B) Would the product be optically active of optically in active?arrow_forwardUnder the required basic conditions, the Suzuki coupling is not efficient when the organic electrophile (RX) is an alkyl halide. What undesired side reaction is possible under basic conditions?arrow_forward
- A B ) Which of the above molecules (A or B) have a higher rate of reaction towards aromatic electrophilic reaction? Explain your answer.arrow_forwardWhich heterocycles are more basic than pyrrole?arrow_forwardName the kind of sigmatropic rearrangement that occurs in each of the following reactions. a. Using arrows, show the electron rearrangement that takes place in each reaction.arrow_forward
- Can you please help me and for a, b, and c, please give a brief explanation of the following rules. Thank you so mucharrow_forwardBromide Bhas normal activity (for a secondary bromide) towards SN1 substitution, but A has much higher reactivity and Chas much lower reactivity.arrow_forwardExplain why compound A will not undergo a ring-opening reaction under thermal conditions, but compound B will.arrow_forward
- Draw the curved arrow mechanisms to obtain the products and comment on the selecitivityarrow_forwardPredict the major organic product of the following reaction for a and or barrow_forwardSince compounds A and B are in different comformations, how do the products of the elimation reaction differ in conformation between these two compounds? Please provide visual drawing to explain.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Alcohols, Ethers, and Epoxides: Crash Course Organic Chemistry #24; Author: Crash Course;https://www.youtube.com/watch?v=j04zMFwDeDU;License: Standard YouTube License, CC-BY