
Concept explainers
What is wrong in each of the following shorthand structures?
a.
b.
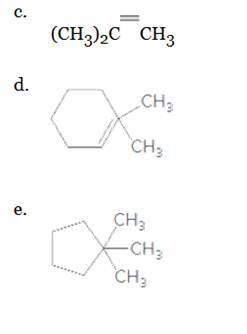
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
CONNECT IA GENERAL ORGANIC&BIO CHEMISTRY
- REPRESENTATIONS OF ORGANIC MOLECULES Identifying isomers and resonance structures Determine the relationship between Structure A and Structure B in each row of the table. Structure A Structure B Relationship H. o isomers H. H H -H H-C- C-O -H- O resonance structures H H. H H Н—С—Н o neither H o isomers H :0: :0-H H. H H- C-C C-H H-C=C C=C-H O resonance structures H. H. H o neither o isomers :o: H H. H- O resonance structures H H H. H o neither 72°F DELL :ö: IIarrow_forwardWhich of the following carbon to carbon bonds has the shortest bond? H. H. H. C=C - C-H - H. H H. H. Option 1 Option 2 Option 3 either A or Barrow_forwardQ#3: Identify A-I and A-D. C,H&O3 1. CH3O 2. CH3Br HO HCI, H2O E Socl, CH3OH 1. CH30 excess H 2. HCI B F excess 1. CH3O 2. CH3B. HCI, H2Oarrow_forward
- a. 2 0=0 H3C CH3 NaOH + 2 H₂Oarrow_forward1 Gmail YouTube Maps Netflix 2.E: Chemical Equili. Chapter 15.3: Solvin. Which of the following structures is NOT an acceptable line formula for CH3CHCICCH? Select one: tof CI O a. H,C CI H. H,C CH O C. All are acceptable. CI d. H,C" CH CI H. O e. H.arrow_forward0 Question 7 Which of the following structure has the R configuration H₂C O b. ll O a. l H 1 O c. I and III -CH₂CH₂ OH O d. I, II and III H₂C H₂CH₂C 11 H H₂C OH H 111 CH₂CH₂arrow_forward
- The chemical structure of ethanol (CH, CH, OH) is shown below. Highlight each atom that is in a methyl group. H H H –C -C О — Н H H :0 :arrow_forwardJict the product of the following organic reaction: CH2-07 CH-O + 3H2 P CH₂-0 Specifically, in the drawing area below, draw the chemical structure of the product P. If there is no produc check the No reaction box under the drawing area. Edit atom or group ם' :0: : :Ö: 0 H C N 0 CI CH CH, CH, NH, NH OH wing a structure. CH,OH NH, P S SH CH₂ H₂C Explanation Check tab 2 3 caps lock A shift W E R S Z alt 5 6 7 8 D F H T ( 2 V B Narrow_forwardacetic acid X X X₁ formic acid methanol X He CH₂COOH(aq) ->> X. He HCOOH(aq) (aq), 8. X₁ He - d. CH₂OH(aq) ->> CH₂(g) + CH₂(g) + CH₂(g) + CO₂(g) CO₂(g) + H₂O(1) CO₂(g) + H₂O(1)arrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
