
Loose Leaf for General, Organic and Biological Chemistry with Connect 2 Year Access Card
4th Edition
ISBN: 9781260269284
Author: Janice Gorzynski Smith Dr.
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 11.2, Problem 11.1PP
Fill in all H's and lone pairs in each compound.
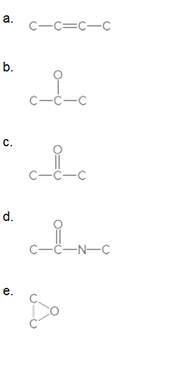
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Match each of the general molecular formulas below with its correct molecular geometry.
Note: The sorting box Tri. Bipyramidal stands for trigonal bipyramidal.
Items (6 items) (Drag and drop into the appropriate area below)
AB5 with no lone
pairs on A
AB4 with one
lone pair on A
AB4 with two
lone pairs on A
AB2 with one
lone pair on A
AB2 with two
AB2 with no lone
lone pairs on A
pairs on A
Categories
Bent 120° bond
Bent 109.5° bond
Tri. bipyramidal
Square planar
Seesaw
Draw the simplest set of curved arrows that shows how the structure on the left could be turned into the structure on the right.
Show all lone pairs. If you need to expand part of the structure to show some lone pairs, expand it by drawing in all atoms and bond lines.
NEC-
N=C=
c*
Predict the indicated bond angles in each compound drawn as a Lewis structure with no implied geometry.
Chapter 11 Solutions
Loose Leaf for General, Organic and Biological Chemistry with Connect 2 Year Access Card
Ch. 11.1 - Prob. 11.1PCh. 11.2 - Fill in all H's and lone pairs in each compound.Ch. 11.3 - Prob. 11.2PPCh. 11.3 - Prob. 11.2PCh. 11.3 - Prob. 11.3PCh. 11.3 - Prob. 11.3PPCh. 11.3 - How many lone pairs are present in lidocaine, the...Ch. 11.4 - Convert each compound to a condensed formula.Ch. 11.4 - Convert each condensed formula to a complete...Ch. 11.4 - Convert each skeletal structure to a complete...
Ch. 11.4 - Prob. 11.5PCh. 11.4 - How many H’s are bonded to each indicated carbon...Ch. 11.4 - Using the skeletal structure, determine the...Ch. 11.5 - Prob. 11.7PCh. 11.5 - Prob. 11.8PCh. 11.5 - For each compound. [1] Identify the functional...Ch. 11.5 - How do a carboxylic acid and an alcohol differ?...Ch. 11.5 - Label each of the following condensed structures...Ch. 11.5 - Prob. 11.11PCh. 11.5 - Prob. 11.12PCh. 11.5 - Identify all of the functional groups in atenolol,...Ch. 11.5 - Prob. 11.13PCh. 11.5 - Prob. 11.10PPCh. 11.5 - Prob. 11.14PCh. 11.6 - Indicate the polar bonds in each compound. Label...Ch. 11.6 - Prob. 11.11PPCh. 11.6 - Prob. 11.16PCh. 11.6 - Predict the water solubility of each compound.Ch. 11.6 - Prob. 11.17PCh. 11.7 - Prob. 11.18PCh. 11.7 - Prob. 11.19PCh. 11.7 - Prob. 11.20PCh. 11 - Prob. 21PCh. 11 - Prob. 22PCh. 11 - Complete each structure by filling in all H’s and...Ch. 11 - Complete the structure of mepivacaine by filling...Ch. 11 - Prob. 25PCh. 11 - Prob. 26PCh. 11 - Prob. 27PCh. 11 - Prob. 28PCh. 11 - “Ecstasy” is a widely used illegal stimulant....Ch. 11 - Prob. 30PCh. 11 - Explain why each C—C—C bond angle in benzene...Ch. 11 - Prob. 32PCh. 11 - Convert each compound to a condensed structure.Ch. 11 - Convert each compound to a condensed structure.Ch. 11 - Convert each compound to a skeletal structure.Ch. 11 - Convert each compound to a skeletal structure.Ch. 11 - Convert each shorthand structure to a complete...Ch. 11 - Convert each shorthand structure to a complete...Ch. 11 - Convert each skeletal structure to a complete...Ch. 11 - Convert each skeletal structure to a complete...Ch. 11 - A and B are ball-and-stick models of two compounds...Ch. 11 - Prob. 42PCh. 11 - What is wrong in each of the following shorthand...Ch. 11 - Prob. 44PCh. 11 - Prob. 45PCh. 11 - Albuterol (trade names Proventil and Ventolin) is...Ch. 11 - Prob. 47PCh. 11 - Prob. 48PCh. 11 - Prob. 49PCh. 11 - (a) Identify the functional groups in donepezil,...Ch. 11 - Prob. 51PCh. 11 - GHB is an addictive, illegal recreational drug...Ch. 11 - Prob. 53PCh. 11 - Prob. 54PCh. 11 - Prob. 55PCh. 11 - Prob. 56PCh. 11 - Prob. 57PCh. 11 - (a) Identify the functional groups in venlafaxine,...Ch. 11 - You are given two unlabeled bottles of solids, one...Ch. 11 - State how potassium iodide (KI) and pentane...Ch. 11 - The given beaker contains 100 mL of the organic...Ch. 11 - Prob. 62PCh. 11 - Why do we need to know the shape of a molecule...Ch. 11 - 1,1-Dichloroethylene (CH2=CCl2) is a starting...Ch. 11 - Indicate the polar bonds in each molecule. Label...Ch. 11 - Indicate the polar bonds in each molecule. Label...Ch. 11 - Classify each molecule as polar or nonpolar.Ch. 11 - Classify each molecule as polar or nonpolar. a....Ch. 11 - Which molecule is more water soluble? Explain.Ch. 11 - Explain why pantothenic acid, vitamin B5, is water...Ch. 11 - Prob. 71PCh. 11 - Prob. 72PCh. 11 - Explain why regularly taking a large excess of a...Ch. 11 - You can obtain the minimum daily requirement of...Ch. 11 - Prob. 75PCh. 11 - Vitamin B6 is obtained by eating a diet that...Ch. 11 - Prob. 77PCh. 11 - Can an oxygen-containing organic compound, have...Ch. 11 - Prob. 79PCh. 11 - Prob. 80PCh. 11 - Benzocaine is the active ingredient in topical...Ch. 11 - Methyl salicylate is responsible for the...Ch. 11 - Answer the following questions about aldosterone,...Ch. 11 - Answer the following questions about...Ch. 11 - Prob. 85PCh. 11 - Skin moisturizers come in two types, (a) One type...Ch. 11 - THC is the active component in marijuana (Section...Ch. 11 - Cocaine is a widely abused, addicting drug....
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 1. For each of the following compounds and ions, draw the complete Lewis structure. All formal charges should be on the atoms that possess that formal charge. CH4 H3CBr CH3CH₂OH H₂S H₂SO4 (sulfuric acid) H³0+ CH3* 2. Convert the following condensed formulas into bond-line structures, making sure to draw in zig-zag formation. CH3CH₂CH₂CH₂CH₂CH₂CH₂CH3 FCH₂CH₂I H₂C=CHCH₂OH (CH3)2CHCOOHarrow_forwardDraw the simplest set of curved arrows that shows how the structure on the left could be turned into the structure on the right. Show all lone pairs. If you need to expand part of the structure to show some lone pairs, expand it by drawing in all atoms and bond lines. c=v` toarrow_forwardDraw VSEPR sketches for each compound below. Then, indicate whether the each of the following compounds is polar or nonpolar by drawing molecular dipole moments when appropriate. CHCl3 CH2Cl2 CCl4 H2O CH3CH2CH2CH2CH3arrow_forward
- Provide a reasonable Lewis structure below each molecular formula. Please note that you must indicate the proper number of lone pairs AlCl3 H3CCH(OH)CH2CO2H H3CCNOarrow_forwardDraw correct line-bond structures for the following moleculesarrow_forwardWrite the letter of the pair of compounds that illustrates the given isomerism. CH, CH CH3 CH OH D в но H,C. H,C. H,C. CH3 Br CHS OH H F CH Br H3C H3C. CH CH CH3 H3C NH2 CH3 K L AND cis-trans geometric isomerism:Iarrow_forward
- draw skeletal structure of this molecule: H2N-CH2-CH-CH2-CH3-CH3arrow_forwardPredict the indicated bond angles in each compound drawn as a Lewis structure with no implied geometry. CH₂-CEC CI: b. CH₂ C CI: c. CH3 CI: Harrow_forwardAlthough we usually don't show lone pairs on skeletal structures, sometimes it's necessary, for example, to show how a reaction will occur. Draw in any lone pairs in the structure below. Note: you may need to redraw parts of the structure to show atoms and bonds within a group before you can add any missing lone pairs ОН OH H,N H,N°arrow_forward
- Predict all bond angles about each highlighted carbon atom. b Blue atom bond angles = Red atom bond angles = H3C -CH₂OH H3C Blue atom bond angles ⒸH₂CC=CC=CH₂ Blue atom bond angles = Red atom bond angles = dH3C. CH3 CH3 Blue atom bond angles =arrow_forwardAlthough we usually don't show lone pairs on skeletal structures, sometimes it's necessary, for example, to show how a reaction will occur. Draw in any lone pairs in the structure below. Note: you may need to redraw parts of the structure to show atoms and bonds within a group before you can add any missing lone pairs c* OH OH NH, NH, toarrow_forwardAre these the same molecule? H H-C- same molecule isomers different molecules -N—H H Harrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning
Types of bonds; Author: Edspira;https://www.youtube.com/watch?v=Jj0V01Arebk;License: Standard YouTube License, CC-BY