
Concept explainers
Interpretation:
To convert the given ball and stick model of 'elemicin' which is a component of nutmeg to skeletal structure and also identify the
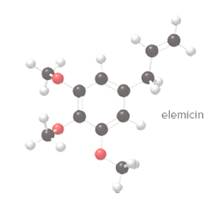
Concept introduction:
Complete structural formula of a molecule represents all the atoms of molecule, types of bonds connecting atoms and how atoms are connected to each other.
Organic molecules have some structural features in addition to
This table gives information about some common functional groups:
| Type of Compound | General Structure |
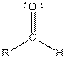 | |
| Ketone | 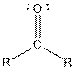 |
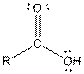 | |
| Ester | 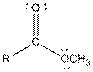 |
| Amide | 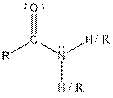 |
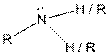 | |
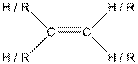 | |
| Ether | 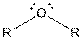 |
| Alcohol | 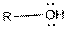 |
Here, R can be any carbon backbone.
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
Loose Leaf for General, Organic and Biological Chemistry with Connect 2 Year Access Card
- Based on the structures below: i) Convert the structures above to the skeletal structure.ii) Identify the functional groups and homologous series in each compound.arrow_forwardWrite condensed structural formulas, and skeletal structures for each structural isomer of C4H10.arrow_forward4: Draw out the abbreviated structural formula of the following molecules. 4-Methyl-2-hexene 2-Methyl-2-hexene 2-Hexene 5: Draw the structure of 2-Butene, labeling the n bond and all the B bonds. 6: Draw all the structures possible with the formula CSH8.arrow_forward
- Classify the organic compounds by the class of their functional group.arrow_forwardClassify the following compounds based on their functional groups. A OH H3C B CH3 H3C CH3 H3C EN H3Carrow_forwardExcluding the aryl group (benzene ring), identify the functional groups present. 1. но NH2 Aspartame Demerolarrow_forward
- encircle and identify the functional groupsarrow_forwardProvide a skeletal line structure for hexanoic acid.arrow_forward(iii) In the presence of a suitable catalyst, 2-methylpropene forms a mixture of dimers. Two of these dimers react with hydrogen to form * 2,2,4-trimethylpentane. Draw the skeletal formula for 2,2,4-trimethylpentane. Use this to draw the skeletal structure of one of the dimers formed from 2-methylpropene. 2,2,4-trimethylpentane Dimerarrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning  ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





