
Concept explainers
Interpretation:
The difference between
Concept introduction:
Organic molecules have some structural features in addition to
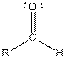
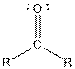
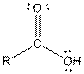
Table given below gives information about some common functional groups:
| Type of Compound | General Structure |
 | |
 | |
 | |
| Ester | 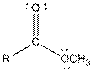 |
| Amide | 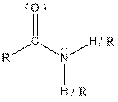 |
| Alcohol |  |
| Amine | 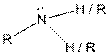 |
 |
Here, R = any carbon backbone
Ball and stick model of molecule is determined by X-ray crystallography, in which the hydrogen atoms are depicted as white balls, carbon atoms as black balls, nitrogen atoms as blue balls and oxygen atom as red balls.
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
Loose Leaf for General, Organic and Biological Chemistry with Connect 2 Year Access Card
- The following molecular formulas represent saturated amine compounds or unsaturated amine compounds that contain C=C bonds. Determine whether each compound is saturated and how many C=C bonds are present. a C,H1N This amine is c and contains C=C bond(s). b C,H12N2 correct lamine is and contains C-C bond(s). € CH,N This amine is and contains C=C bond(s).arrow_forwardIs the substituted group in NAG an amine or an amide? There is a carbon double bonded to an oxygen, and also attached to a nitrogen, however, the PPT says it is an amine.arrow_forwardHow do the neurotransmitters serotonin and histamine vary in structures.arrow_forward
- acetylsalicylic acid vs salicylic acid? What is the main chemical difference? Why can acetylsalicylic acid pass through the stomach without harm while salicylic acid can cause harm to the stomach????arrow_forwardно What kind of amine is found in this compound? What kind of carbon atom is the OH group attached to in this compound? What is the name of the carbon group with a box around it? Nearrow_forwardWhat type of amine is this? 'N'arrow_forward
- amine, (2) an amide, or (3) both an amine and an amide. 17-106 Classify each of the following compounds as (1) an amine, (2) an amide, or (3) both an amine and an NH2 b. `NH a. H2N H d. с.arrow_forwardWrite the names for the amines using the naming styles taught in the McMurry text. H2 H3C-N-C-CH3 name: H2 H2 H3C-C-Ņ-c-CH3 H name: These compounds are amines.arrow_forwardA) Name the structure B) Can this amine form an amide? If yes, show below the structure of the amide formed with acetic acid. If no, tell why an amide cannot be formed.arrow_forward
- 2. What is produced when an amine reacts with water? A. A primary alcohol and ammonia B. An amide and a hydrogen (H+) ion C. An ammonium ion and a hydroxide (OH-) ion D. An amide and a hydroxide (OH-) ionarrow_forward2. What is produced when an amine reacts with a strong acid such as HCl? A. An amine and the OH- ion B. An amide and the H+ ion C. An ammonium hydroxide D. An ammonium saltarrow_forwardAmide hydrolysis in basic conditions forms A. a carboxylic acid and an amine B. a carboxylate salt and an amine 3. an ester and an amine 4. a carboxylic acid and an amine saltarrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
