
Organic Chemistry, Loose-leaf Version
8th Edition
ISBN: 9781305865549
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 11.4, Problem 11.3P
Show how you might use the
(a) 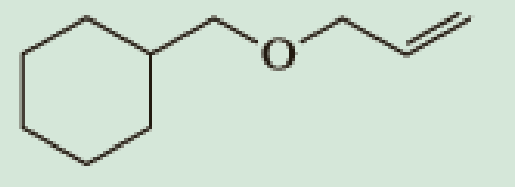
(b) 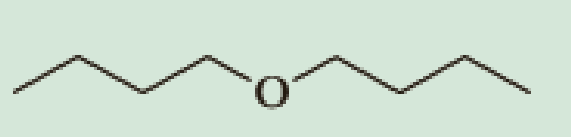
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Which ether will not easily react with concentrated HI?
(a)
(b)
(c)
(d)
Provide reagents
Draw all constitutional isomeric ethers with the molecular formula C4H10O. Provide a common name and a systematic name for each isomer.
Chapter 11 Solutions
Organic Chemistry, Loose-leaf Version
Ch. 11.2 - Write IUPAC and common names for these ethers. (a)...Ch. 11.3 - Arrange these compounds in order of increasing...Ch. 11.4 - Show how you might use the Williamson ether...Ch. 11.4 - Show how ethyl hexyl ether might be prepared by a...Ch. 11.5 - Account for the fact that treatment of tert-butyl...Ch. 11.5 - Draw structural formulas for the major products of...Ch. 11.6 - Prob. 11.7PCh. 11.8 - Draw the expected products of Sharpless...Ch. 11.9 - Prob. AQCh. 11.9 - Prob. BQ
Ch. 11.9 - Prob. CQCh. 11.9 - Prob. DQCh. 11 - Write names for these compounds. Where possible,...Ch. 11 - Prob. 11.11PCh. 11 - Each compound given in this problem is a common...Ch. 11 - Account for the fact that tetrahydrofuran (THF) is...Ch. 11 - Prob. 11.14PCh. 11 - Write equations to show a combination of reactants...Ch. 11 - Propose a mechanism for this reaction.Ch. 11 - Prob. 11.17PCh. 11 - Prob. 11.18PCh. 11 - Prob. 11.19PCh. 11 - Prob. 11.20PCh. 11 - Ethylene oxide is the starting material for the...Ch. 11 - Prob. 11.22PCh. 11 - Predict the structural formula of the major...Ch. 11 - The following equation shows the reaction of...Ch. 11 - Propose a mechanism to account for this...Ch. 11 - Acid-catalyzed hydrolysis of the following epoxide...Ch. 11 - Prob. 11.27PCh. 11 - Prob. 11.28PCh. 11 - Prob. 11.29PCh. 11 - Propose a mechanism for the following...Ch. 11 - Show reagents and experimental conditions to...Ch. 11 - Starting with cis-3-hexene, show how to prepare...Ch. 11 - Show reagents to convert cycloheptene to each of...Ch. 11 - Show reagents to convert bromocyclopentane to each...Ch. 11 - Prob. 11.35PCh. 11 - Starting with acetylene and ethylene oxide as the...Ch. 11 - Following are the steps in the industrial...Ch. 11 - Prob. 11.38PCh. 11 - Prob. 11.39PCh. 11 - Aldehydes and ketones react with one molecule of...Ch. 11 - Prob. 11.42PCh. 11 - Write the products of the following sequences of...Ch. 11 - Using your reaction roadmap as a guide, show how...Ch. 11 - Using your reaction roadmap as a guide, show how...Ch. 11 - Using your reaction roadmap as a guide, show how...Ch. 11 - During the synthesis of the antiasthmatic drug...Ch. 11 - Prob. 11.48P
Additional Science Textbook Solutions
Find more solutions based on key concepts
Classify each example of molecular art as a pure element, a pure compound, or a mixture.
General, Organic, and Biological Chemistry - 4th edition
Q2. Which statement best defines chemistry?
a. The science that studies solvents, drugs, and insecticides
b. Th...
Introductory Chemistry (5th Edition) (Standalone Book)
Write the electron configurations far each of the following elements: (a) Sc. (b) Ti. (c) Cr. (d) Fe. (e) Ru
Chemistry by OpenStax (2015-05-04)
The active ingredient in Tylenol and a host of other over-the-counter pain relievers is acetaminophen (C8H9NO2)...
Chemistry: Atoms First
What is the pH range for acidic solutions? For basic solutions?
Introduction to Chemistry
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Phenylethanol can be oxidised to phenylethanal or phenylethanoic acid, depending on the reagents used (both the alcohol and the aldehyde are of interest for their antimicrobial properties, while the acid is used to treat type II hyperammonemia): A (a) (b) (c) CoH,CH,CHO phenylethanal B C6H5CH₂CH₂OHC₂H₂CH₂CO₂H phenylethanol Suggest reagents (shown as A and B in the scheme above) that could be used to carry out the oxidation of the alcohol to the aldehyde and the acid, respectively. C6H5CH₂- Suggest two other syntheses of phenylethanoic acid, in each case indicating the starting materials and other reagents required, but not giving details of mechanism. One of your proposed syntheses must start with a compound which only contains seven carbon atoms (the acid product contains eight carbon atoms). phenylethanoic acid Phenylethanal can be converted to a hydrate in the presence of aqueous acid, though the position of equilibrium is very far to the left: H H+/H₂O OH C6H5CH₂-C-H OH Explain why…arrow_forward(c) Draw the structure of the hemi-acetal and acetal formed when cyclohexanone is combined with methanol in the presence of catalytic acid.arrow_forwardRank compound A, B, and C in the order of increasing stability.arrow_forward
- Acyclovir is an effective antiviral agent used to treat the herpes simplexvirus. (a) Draw the enol form of acyclovir, and explain why it is aromatic.(b) Why is acyclovir typically drawn in its keto form, despite the fact thatits enol is aromatic?arrow_forwardAcyclovir is an effective antiviral agent used to treat the herpes simplex virus. (a) Draw the enol form of acyclovir, and explain why it is aromatic. (b) Why is acyclovir typically drawn in its keto form, despite the fact that its enol is aromatic?arrow_forward16.48 D is an intermediate in the synthesis of rosiglitazone (trade name Avandia), a drug used to treat type 2 diabetes. Suggest two different methods to prepare the ether in D by substitution reactions. CHO D omoss rosiglitazone Harrow_forward
- Avobenzone is a conjugated compound that absorbs ultraviolet light with wavelengths in the 320–400 nm region, so it is a common ingredient in commercial sunscreens. Write out two different crossed Claisen reactions that form avobenzone.arrow_forwardDraw a stepwise mechanism for the following reaction that forms ether D. D can be converted to the antidepressant fluoxetine (trade name Prozac) in a single step.arrow_forward(a) What is the major alkene formed when A is dehydrated with H2SO4? (b) What is the major alkene formed when A is treated with POCl3 and pyridine? Explain why the major product is different in these reactions.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY