
Organic Chemistry, Loose-leaf Version
8th Edition
ISBN: 9781305865549
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Chapter 11.9, Problem BQ
Interpretation Introduction
Interpretation:
The product distribution that tells a chemist about the mechanism of the acid-catalyzed epoxide ring opening has to be discussed.
Concept Introduction:
Stereochemistry: The spatial arrangement of atoms or groups present in compound.
Ring opening: Ring opening of epoxide takes place by acid-catalyzed hydrolysis of an epoxide.
Acid-catalyzed hydrolysis of an epoxide:
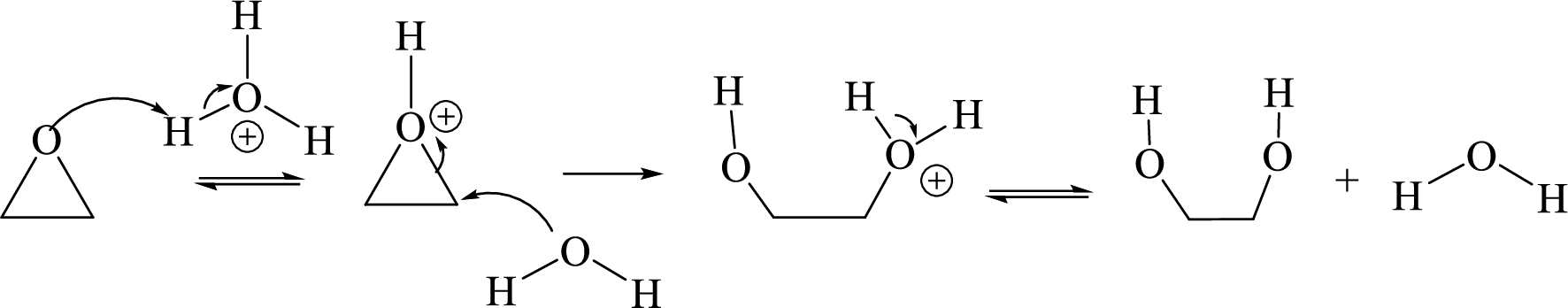
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
When performing a nucleophilic substitution reaction and not knowing the structure of the starting material or product, how can you form a method to determine whether the reaction proceeds via an SN1 or SN2 mechanism?
Please explain the steps of this synthesis. Identify SN1, SN2, E2 or E1. Identify the nucleophile and electrophiles
22. Which of the following statements is generally true for SN1 reactions?
A) Complete inversion of configuration occurs.
B) These are favored by nonpolar solvents.
C)These reactions are favored by polar solvents.
D) Reaction rates depend only on the concentration of the nucleophile.
E) The mechanism is a one-step back attack.
23. Which of the alkyl chlorides listed below undergoes dehydrohalogenation in the presence of a strong
base to give 2-pentene as the only alkene product?
A) 1-chloropentane
B) 2-chloropentane
C) 3-chloropentane
D) 1-chloro-2-methylbutane
E) 1-chloro-3-methylbutane
Chapter 11 Solutions
Organic Chemistry, Loose-leaf Version
Ch. 11.2 - Write IUPAC and common names for these ethers. (a)...Ch. 11.3 - Arrange these compounds in order of increasing...Ch. 11.4 - Show how you might use the Williamson ether...Ch. 11.4 - Show how ethyl hexyl ether might be prepared by a...Ch. 11.5 - Account for the fact that treatment of tert-butyl...Ch. 11.5 - Draw structural formulas for the major products of...Ch. 11.6 - Prob. 11.7PCh. 11.8 - Draw the expected products of Sharpless...Ch. 11.9 - Prob. AQCh. 11.9 - Prob. BQ
Ch. 11.9 - Prob. CQCh. 11.9 - Prob. DQCh. 11 - Write names for these compounds. Where possible,...Ch. 11 - Prob. 11.11PCh. 11 - Each compound given in this problem is a common...Ch. 11 - Account for the fact that tetrahydrofuran (THF) is...Ch. 11 - Prob. 11.14PCh. 11 - Write equations to show a combination of reactants...Ch. 11 - Propose a mechanism for this reaction.Ch. 11 - Prob. 11.17PCh. 11 - Prob. 11.18PCh. 11 - Prob. 11.19PCh. 11 - Prob. 11.20PCh. 11 - Ethylene oxide is the starting material for the...Ch. 11 - Prob. 11.22PCh. 11 - Predict the structural formula of the major...Ch. 11 - The following equation shows the reaction of...Ch. 11 - Propose a mechanism to account for this...Ch. 11 - Acid-catalyzed hydrolysis of the following epoxide...Ch. 11 - Prob. 11.27PCh. 11 - Prob. 11.28PCh. 11 - Prob. 11.29PCh. 11 - Propose a mechanism for the following...Ch. 11 - Show reagents and experimental conditions to...Ch. 11 - Starting with cis-3-hexene, show how to prepare...Ch. 11 - Show reagents to convert cycloheptene to each of...Ch. 11 - Show reagents to convert bromocyclopentane to each...Ch. 11 - Prob. 11.35PCh. 11 - Starting with acetylene and ethylene oxide as the...Ch. 11 - Following are the steps in the industrial...Ch. 11 - Prob. 11.38PCh. 11 - Prob. 11.39PCh. 11 - Aldehydes and ketones react with one molecule of...Ch. 11 - Prob. 11.42PCh. 11 - Write the products of the following sequences of...Ch. 11 - Using your reaction roadmap as a guide, show how...Ch. 11 - Using your reaction roadmap as a guide, show how...Ch. 11 - Using your reaction roadmap as a guide, show how...Ch. 11 - During the synthesis of the antiasthmatic drug...Ch. 11 - Prob. 11.48P
Knowledge Booster
Similar questions
- 22. Which of the following statements is generally true for SN1 reactions? A) Complete inversion of configuration occurs. B) These are favored by nonpolar solvents. C)These reactions are favored by polar solvents. D) Reaction rates depend only on the concentration of the nucleophile. E) The mechanism is a one-step back attack. 23. Which of the alkyl chlorides listed below undergoes dehydrohalogenation in the presence of a strong base to give 2-pentene as the only alkene product? A) 1-chloropentane B) 2-chloropentane C) 3-chloropentane D) 1-chloro-2-methylbutane E) 1-chloro-3-methylbutane 24. Which of the following statements correctly describe(s) El reactions of alkyl halides (RX)? I. Rate = k[base] II. Rate = k[base][RX] III. Rate = K[RX] IV. The reactions occur in two distinct steps. Rearrangements are sometimes seen. V. A) II and IV B) III and V C) I, IV, and V D) I only E) III, IV, and Varrow_forwardNucleophilic aromatic substitution involves the formation of a resonance-stabilized carbanion intermediate called a Meisenheimer complex as the nucleophile attacks the ring carbon carrying the eventual leaving group; electron-withdrawing groups ortho and/or para to the site of attack help to stabilize this structure via resonance. For the reaction below, draw the structure of the stabilized reaction intermediate in the box below. CI F3C CF3 NO₂ F3C N CF3 NO₂ • You do not have to consider stereochemistry. • Draw the Meisenheimer complex with a formal charge of 0 on the nucleophilic atom. If more than one resonance structure is possible, only draw the most important one.arrow_forward22. a. Label the reactive features, highlight the most reactive one, then highlight what it needs. Also, state if the reaction will start to create a carbocation, carbon radical, or carbanion, or will cause loss of aromatic character. If a carbocation, carbon radical, or carbanion starts to develop, label where that will occur. sec-butyl ethyl ether + HCl in water b. Use mechanism arrows to illustrate the reaction that occurs. c. If applicable, use stabilization resources to deal with the carbocation, carbon radical, or carbanion that starts to develop during the reaction, and draw the structure of any resonance-stabilized intermediate. d. Continue labeling and diagramming the reaction until you find the major stable product(s). e. Finally, state the stereochemistry of the major product(s) and use either Fisher projection or perspective formula representations to illustrate that stereochemistry.arrow_forward
- Give the products of the following substitution reactions. For every reaction, show electron pairs on both nucleophile and leaving group.arrow_forwardThe SN1 mechanism starts with the rate-determining step which is the dissociation of the alkyl halide into a carbocation and a halide ion. The next step is the rapid reaction of the carbocation intermediate with the nucleophile; this step completes the nucleophilic substitution stage. The step that follows the nucleophilic substitution is a fast acid-base reaction. The nucleophile now acts as a base to remove the proton from the oxonium ion from the previous step, to give the observed product. Draw a curved arrow mechanism for the reaction, adding steps as necessary. Be sure to include all nonzero formal charges.arrow_forward1. Predict the product, and propose a mechanism for the following nucleophilic addition to aldehyde or ketone. Are the products different from one another? Why do the reactions proceed via different mechanisms? Note the charges on the intermediates of acid and base catalyzed reactions. Oxygen nucleophile in acidic condition H2O, [H+] Oxygen nucleophile in basic condition NaOHarrow_forward
- Choose the reagent(s) that would be most likely to complete this reaction. I||| / A B C D 1. BH3-THF 2. H2O2, NaOH Br₂ H₂O OsO4 (catalytic) NMO RCO3H Donearrow_forwardThe following reaction gives a mixture of the nucleophilic substitution (SN1 or SN2) and elimination (E1 or E2) products. Propose two mechanisms that can account for the formation of these products. One mechanism should be for the nucleophilic substitution reaction and the other mechanism would show the elimination reaction.arrow_forwardwhat is the product and mechanism used in this reaction?arrow_forward
- In a reaction between a certain alkyl halide and sodium cyanide, it was found that decreasing the alkyl halide concentration by one half, the rate of the reaction decreased by one half. Which of the following is a reasonable conclusion based on this information? b) The reaction proceeds by the E1 mechanism. c) The reaction proceeds by the SN2 mechanism. a) The reaction proceeds by the SN1 mechanism. d) The reaction proceeds by the E2 mechanism. e) None of these is a reasonable conclusion.arrow_forwardExplain why a methoxy group (CH3O) increases the rate of electrophilic aromatic substitution, but decreases the rate of nucleophilic aromatic substitution.arrow_forwardRank these substrates in order of increasing ability to undergo a nucleophilic substitution aromatic (SNAr) reaction.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning