
Concept explainers
(a)
Interpretation:
The reason should be explained for difficulty in the synthesis of unsymmetrical ether.
Concept introduction:
Ethers:
An oxygen atom connected to two alkyl or aryl groups is called as ether. The general formula of ether is
Ethyl alcohol is reaction with acid which yields diethyl ether which is shown below.
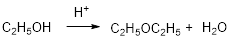
(b)
Interpretation:
The ethyl propyl ether should be synthesized.
Concept introduction:
Ethers:
An oxygen atom connected to two alkyl or aryl groups is called as ether. The general formula of ether is
Ethyl alcohol is reaction with acid which yields diethyl ether which is shown below.
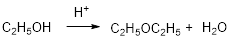
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
Organic Chemistry; Modified MasteringChemistry with Pearson eText -- ValuePack Access Card; Study Guide and Student Solutions Manual for Organic Chemistry, Books a la Carte Edition (7th Edition)
- What is the name for this compound? Select one: a. 2-bromo-5-ethylphenol b. 4-bromo-1-ethyl-5-phenol c. 6-bromo-3-ethylphenol d. 2-bromo-5-ethylcyclohexanol e. 2-bromo-5-methylphenolarrow_forwardWhat is produced when methanal undergoes Grignard reaction? O A. primary alcohol B. secondary alcohol O C. tertiary alcohol O D. no reactionarrow_forward1. An alkene reacts with water with an acid catalyst results into a formation of: A. Aldehyde B. Ketone C. Alcohol D. Ester 2. 3-Methylhexanal with K2Cr2O7 will yield: A. 3-Methyl-1-hexanol B. 3-Methylhexanoic acid C. 3-Methyl-1-hexanone D. 3-Methyl-1-hexanethiol 3. This is a reverse process of Hydration reaction: A. Oxidation reaction B. Reduction reaction C. Dehydration reaction D. Hydration reaction 4. Acetic acid reacts with a strong base forms: A. Salt B. Water C. Salt and Water D. No reaction 5. Ketones can be further oxidized with benedict's solution into: A. Alcohol B. Aldehyde C. Catalysts D. No reactionarrow_forward
- 1. Cite some reactions in which formaldehyde behaves differently from other aldehydes. 2. Give some biological and medical applications of: a. formalin b. chloral c. urotropinearrow_forward1. Match the following: A. alcohol (phenol) B. Ether CH3-CH2-CHO СН3-СН2-С-СНЗ C. Amine С6Н5-СООН D. aldehyde С2H5-СООСHЗ E. Ketone F. Thiol G. carboxylic acid С6Н5-ОН СНЗ-О-С2Н5 CH3-SH H. Ester C2H5-NH2 I. Amide H-C-NH2 2. Name the following: CH3 CH3-C -CH2- Č -CH3 CH3 CH3 CH3 CH3 CH3-C-CH3 CH3 – C- C=C - C6H5arrow_forward1. What are the substances present in the label of formaldehyde (formalin)? 2. What is the percentage composition of acetone in the nail polish remover? 3. According to your interview, what is/are the uses of formalin and acetone? A. Uses of Formalin- B. Uses of Acetone - 4. What type of bond is present in the two structures? 5. Why do you think aldehydes and ketones belong to the group of carbonyl containing compounds?arrow_forward
- The sub-part to be solved... 4. Acetic acid reacts with a strong base forms: A. Salt B. Water C. Salt and Water D. No reaction 5. Ketones can be further oxidized with benedict's solution into: A. Alcohol B. Aldehyde C. Catalysts D. No reactionarrow_forwardWhich alcohol undergoes dehydration fastest ? O a. l-methylcyclohexanol O b. 1-pentanol O c.n-butyl alcohol O d. iso-butyl alcohol O e. cyclopentanolarrow_forward1. Draw the following compounds. c. (Z)-2-methylpent-2-enol d. ethylpropanoatearrow_forward
- Compare aldehydes and ketones as to: (Use acetaldehyde and acetone as examples). A. Reaction with cone. NaOH, heated B. Reaction with phenylhydrazinearrow_forwardWhich among the following reagent in the choices can be utilized to differentiate propionaldehyde from ethyl propyl ketone? A. Schiff’s reagent B. 2,4-DNPH C. Alcoholic Silver Nitrate D. Sodium bisulfate E. None of the above Kindly explain your answer.arrow_forwardFLAVORANTS: Identify the aldehyde and ketone-containing flavoring compound from each following sources. Choose your best answer from the choices below A. 2-Octanone B. Citral C. Benzaldehyde D. Vanillin E. Cinnamaldehyde F. a-Damascone 1. Вerry 2. Mushroom 3. Lemongrass 4. Cinnamon 5. Almonds Idchyde or ketone formed froarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





