
ORGANIC CHEMISTRY(LL)-W/SG.+SOLN+ACCESS
8th Edition
ISBN: 9780134649818
Author: Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Question
Chapter 11.4, Problem 15P
Interpretation Introduction
Interpretation:
The hydrocarbon used to prepare the Organoborane compound given below has to be identified.
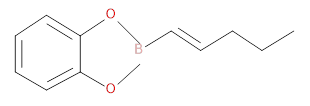
Concept introduction:
Hydrocarbon:
A hydrocarbon is an organic compound consisting entirely of hydrogen and carbon .
Hydrogen is examples of group 14 hydrides.
Hydride ion:
Negatively charged hydrogen (a hydrogen atom with an extra electron).
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What is the acid anhydride of phosphoric acid (H3PO4)?
16.
How many moles of H2O would be produced by the complete combustion of 1.0 mole of cyclobutane?
a. 3.0
b. 4.0
c. 5.0
d. 6.0
What reagent serves as the source of ammonia to deprotonate oxalic acid in the production of calcium oxalate monohydrate?
a. ammonium oxalate
b. hydrochloric acid
c. urea
d. ammonium hydroxide
Chapter 11 Solutions
ORGANIC CHEMISTRY(LL)-W/SG.+SOLN+ACCESS
Ch. 11.1 - Prob. 1PCh. 11.2 - Which is more reactive an organolithium compound...Ch. 11.2 - Prob. 3PCh. 11.3 - Muscalure is the sex attractant of the common...Ch. 11.3 - Prob. 7PCh. 11.3 - Prob. 8PCh. 11.3 - Prob. 9PCh. 11.3 - Prob. 10PCh. 11.4 - Prob. 13PCh. 11.4 - Prob. 14P
Ch. 11.4 - Prob. 15PCh. 11.4 - Prob. 16PCh. 11.4 - Prob. 17PCh. 11.4 - Prob. 19PCh. 11.4 - Show how the Suzuki and/or Heck reactions can be...Ch. 11.4 - Identify two pairs of an alkyl bromide and an...Ch. 11.5 - Prob. 22PCh. 11.5 - Draw the product of ring-closing metathesis for...Ch. 11.5 - Prob. 25PCh. 11.5 - Prob. 26PCh. 11 - Prob. 27PCh. 11 - Prob. 28PCh. 11 - The coupling of an alkyne with an aryl halide in...Ch. 11 - Identify A through H.Ch. 11 - Using the given starting material, any necessary...Ch. 11 - What alkyl halide reacts with lithium...Ch. 11 - Prob. 33PCh. 11 - Prob. 34PCh. 11 - The following compound undergoes an intramolecular...Ch. 11 - Using ethynyleyclohexane as a starting material...Ch. 11 - Prob. 37PCh. 11 - Using the given starting material, any necessary...Ch. 11 - Prob. 39PCh. 11 - A student added an equivalent of...Ch. 11 - Using the given starting material, any necessary...Ch. 11 - Prob. 42PCh. 11 - Prob. 43PCh. 11 - Bombykol is the sex pheromone of the silk moth....Ch. 11 - Prob. 45PCh. 11 - Prob. 46PCh. 11 - A dibromide loses only one bromine when it reacts...Ch. 11 - What starting material is required in order to...Ch. 11 - What product is obtained from ring-opening...
Knowledge Booster
Similar questions
- Why are the interactions between H2O molecules stronger than those between HF molecules, even though the polarity of the HF bond is larger than the polarity of the OH bond?arrow_forwardwhich molecule in the reaction below undergoes oxidation? 4Fe + 3 O2 > 2Fe2O3arrow_forwardwrite the reactions in which one of the following compounds acts on the ammonia solution of silver oxide. a) propene b) bytine-2 c) cyclopropane d) pentin-1arrow_forward
- (a) Name each of the following compounds and its functional group. (1) CH3CH2 CH2OH (2) CH3CH2NH2 (b) Name the product from the reaction of compounds 1 and 2 above. (c) What is the name and structure of the product from the oxidation of 1 with an excess of oxidizing agent? (d) Give the name and structure of the compound that results from combining 2 and 3. (e) What is the result of adding an acid (say HCl) to compound 3?arrow_forwardCan you pick the correct answer in each bracket In step 8, starting compound undergoes [ oxidation / hydrolysis / reduction] with [PCC / DCC / NaOH or H3O+] to form (8).arrow_forwardDraw the structure of the predominant form of CF3CH2OH (pK a = 12.4) at pH = 6.arrow_forward
- Write balanced chemical equations for the reaction of the following acid anhydrides with water: (a) SO3 (b) N2O3arrow_forwardfind out which compound acts on hydrogen chloride, bromine, ammonia solution of Ag + .Write the corresponding reactions. a) Butene-2 b) chloromethane c) butyne -2 d) propinearrow_forwardhow H2O2 can be prepared on the large scale?arrow_forward
- Determine the direction of equilibrium when acetylene (HC≡CH) reacts with−NH2 in a proton transfer reaction ?arrow_forwardWhat is the potential effect of Terephthalic acid synthesis on human life and the environment?arrow_forwardThis is a reduction reaction. Two hydrogens and two electrons are used to reduce the ketone to an alcohol. Draw out the structures of NaBD4 and EtOH. What is their role in this reaction? Draw the product of the reaction. What advantage is there to using these reducing agents instead of hydrogenation reagents (H2, metal catalyst)?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning  Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
