
CHEMISTRY:INTRO.GEN ORG.W/MODIFIED MAS
13th Edition
ISBN: 9780134809649
Author: Timberlake
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 11.7, Problem 11.34PP
Draw the structural formula for the product in each of the following reactions:
a. CH3 CH2 CH2 -CH = CH2+H20
b.
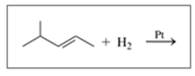
c.
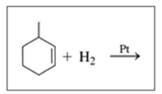
d.
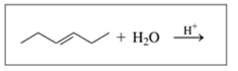
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
When synthesizing acetals and ketals, what is the main driving force behind whether the reaction favors the carbonyl species or the acetal/ketal?
a the catalytic acid
b solvent
c temperature
d Le Châtelier's Principle
Draw the product(s) of the reaction between 1-propyne (1 mole) and the reagents given below.
A) 2 moles HBr
B) excess H2 over Pd-C
C) excess H2 in Lindlar’s catalyst
Claisen Condensation:
7.50 grams of methyl acetate is reacted with water and sulfuric acid catalyst under heated conditions. What amount of water (in grams) is required for the reaction and what is the theoretical yield of the acetic acid product? Include a balanced equation.
Chapter 11 Solutions
CHEMISTRY:INTRO.GEN ORG.W/MODIFIED MAS
Ch. 11.1 - Identify each of the following as a formula of an...Ch. 11.1 - Identify each of the following as a formula of an...Ch. 11.1 - Identify each of the following properties as more...Ch. 11.1 - Identify each of the following properties as more...Ch. 11.1 - Prob. 11.5PPCh. 11.1 - Prob. 11.6PPCh. 11.2 - Give the IUPAC name for each of the following...Ch. 11.2 - Give the IUPAC name for each of the following...Ch. 11.2 - Draw the condensed structural formula for alkanes...Ch. 11.2 - Draw the condensed structural formula for alkanes...
Ch. 11.3 - Indicate whether each of the following pairs...Ch. 11.3 - Indicate whether each of the following pairs...Ch. 11.3 - Give the IUPAC name for each of the following a....Ch. 11.3 - Give the TUPAC name for each of the following: a....Ch. 11.3 - Draw the condensed structural formula for each of...Ch. 11.3 - Draw the condensed structural formula for each of...Ch. 11.3 - Draw the line-angle formula for each of the...Ch. 11.3 - Prob. 11.18PPCh. 11.4 - Heptane, used as a solvent for rubber cement, has...Ch. 11.4 - Nonane has a density of 0.79 g/mL and boils at 151...Ch. 11.4 - Write the balanced chemical equation for the...Ch. 11.4 - Write the balanced chemical equation for the...Ch. 11.5 - Identify the following as alkanes, alkenes,...Ch. 11.5 - Identify the following as alkanes, alkenes,...Ch. 11.5 - Give the IUPAC name for each of the following: a....Ch. 11.5 - Give the IUPAC name for each of the following: a....Ch. 11.5 - Draw the condensed structural formula, or...Ch. 11.5 - Prob. 11.28PPCh. 11.6 - Give the IUPAC name for each of the following,...Ch. 11.6 - Give the IUPAC name for each of the following,...Ch. 11.6 - Draw the condensed structural formula for each of...Ch. 11.6 - Prob. 11.32PPCh. 11.7 - Draw the structural formula for the product in...Ch. 11.7 - Draw the structural formula for the product in...Ch. 11.8 - Give the IUPAC name for each of the following: a....Ch. 11.8 - Give the IUPAC name for each of the following: a....Ch. 11.8 - Draw the line-angle formula for each of the...Ch. 11.8 - Draw the line-angle formula for each of the...Ch. 11.8 - Write the balanced chemical equation for the...Ch. 11.8 - Write the balanced chemical equation for the...Ch. 11 - Prob. 11.41UTCCh. 11 - Prob. 11.42UTCCh. 11 - Prob. 11.43UTCCh. 11 - Prob. 11.44UTCCh. 11 - Convert each of the following line-angle formulas...Ch. 11 - Convert each of the following line-angle formulas...Ch. 11 - Give the IUPAC name for each of the following:...Ch. 11 - Give the IUPAC name for each of the following:...Ch. 11 - Give the IUPAC name (including cis or trans, if...Ch. 11 - Give the LUPAC name (including cis or trans, if...Ch. 11 - Prob. 11.51APPCh. 11 - Prob. 11.52APPCh. 11 - Name each of the following aromatic compounds:...Ch. 11 - Prob. 11.54APPCh. 11 - Draw the condensed structural or line-angle...Ch. 11 - Draw the condensed structural or line-angle...Ch. 11 - Draw the cis and trans isomers for each of the...Ch. 11 - Draw the cis and trans isomers for each of the...Ch. 11 - Prob. 11.59APPCh. 11 - Draw the line-angle formula for each of the...Ch. 11 - Write a balanced chemical equation for the...Ch. 11 - Write a balanced chemical equation for the...Ch. 11 - Give the name for the product from the...Ch. 11 - Give the name for the product from the...Ch. 11 - Draw the condensed structural or line-angle...Ch. 11 - Draw the condensed structural or line-angle...Ch. 11 - Prob. 11.67CPCh. 11 - Prob. 11.68CPCh. 11 - Prob. 11.69CPCh. 11 - Prob. 11.70CPCh. 11 - Prob. 11.71CPCh. 11 - Prob. 11.72CPCh. 11 - Prob. 11.73CPCh. 11 - Margarines are produced from the hydrogenation of...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw structural formulas for the major products of each reaction. (a) (b)arrow_forwardIzopropanole doesn't form by Select one: a. Hydration of propene b. Reduction of propanal c. Hydration of 2-chloropropane d. Reduction of propan-2-onearrow_forwardWhat is the limiting reagent, and their ratios to each other? ( example: 2:3:1) Bromobenzene: 4.5mL Magnesium:1.0g Methyl benzoate:2.5mL Product: 1.82garrow_forward
- Draw a structural formula of the major alkene formed in each b-elimination.arrow_forwardPhosgene (COCl2) was used as a poison gas in World War I. What product would be formed from the reaction of phosgene with each of the followingreagents?a. one equivalent of methanol b. excess methanol c. excess propylamine d. excess waterarrow_forwardBased on the following samples, which is most readily oxidized? a. n-butyl alcohol b. sec-butyl alcohol c. tert-butyl alcohol d. Acetaldehyde e. Benzaldehyde f. Acetone g. Acetophenone h. Isopropyl alcoholarrow_forward
- 11. The stabilized ether contains a stadilizer-antioxidant?A) Methyl alcohol in a concentration of not more than 0.1%B) Para-phenylenediamineC) Resorcinol not less than 0.1%D) Thymol not less than 0.1%arrow_forward1. Enumerate the classes of hydrocarbons that are oxidizable by Baeyer’s reagent. 2. What is the structural feature of these classes of hydrocarbons? 3. Reactions with Baeyer's reagent: a. Cyclohexane b. Naphthalene c. Hexane d. Corn oil e. Heptane f. Ethynearrow_forward#20 B Draw structural formulas for all possible carbocations formed by the reaction of each alkene with HCl.arrow_forward
- Identify the products A and B formed in the following reaction :(a) CH3— CH2— CH = CH — CH3 + HCl → A + Barrow_forwardOxidation of 1-butanol and 2-butanol yields (respectively) a. butanal exclusively b. 2-butanone exclusively c. butanal and 2-butanone d. butanal and 2-butanone if PCC is the oxidizing agent e. butanal and 2-butanone if the Jones reagent is the oxidizing agentarrow_forwardComplete each of the following reactions: a. b.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

07 Physical Properties of Organic Compounds; Author: Mindset;https://www.youtube.com/watch?v=UjlSgwq4w6U;License: Standard YouTube License, CC-BY