
a)
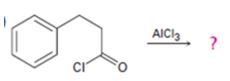
Interpretation:
The products formed in the reaction given are to be identified. The mechanism of the reaction also is to be provided.
Concept introduction:
The mechanism of the electrophilic substitution reaction is to be given. In the first step the electrophile is produced. In the second step the electrons of the
To identify:
The products formed in the reaction given and to provide the mechanism of the reaction.
b)
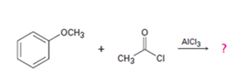
Interpretation:
The products formed in the reaction given are to be identified. The mechanism of the reaction also is to be provided.
Concept introduction:
The mechanism of the electrophilic substitution reaction is to be given. In the first step the electrophile is produced. In the second step the electrons of the aromatic ring attacks the elctrophile to give a resonance stabilized intermediate. In the last step the intermediate deprotonates to yield the product. The methoxyl group is an ortho and para orienting group.
To identify:
The products formed in the reaction given and to provide the mechanism of the reaction.
c)
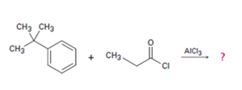
Interpretation:
The products formed in the reaction given are to be identified. The mechanism of the reaction also is to berovided.
Concept introduction:
The mechanism of the electrophilic substitution reaction is to be given. In the first step the electrophile is produced. In the second step the electrons of the aromatic ring attacks the elctrophile to give a resonance stabilized intermediate. In the last step the intermediate deprotonates to yield the product.
To identify:
The products formed in the reaction given and to provide the mechanism of the reaction.
Trending nowThis is a popular solution!

Chapter 11 Solutions
Organic Chemistry
- Complete the following reactions, write major product(s) and specify the mechanism if; SN1 SN2, E1, or E2arrow_forwardWrite the major product of each reaction and what kind of mechanism it is: sn1, sn2, e1, e2.arrow_forwardBased on your knowledge in biological chemistry reaction in biomolecules, what do you think is the best type and product for the above reaction? a. SN1 b. SN2 c. E1 d. E2 e. None of the abovearrow_forward
- Classify the following reaction as an Sn1, Sn2, E1, E1cB, E2 reaction. Can you show me the leaving group and explain how to find a leaving group? And how to identify the other reactions?arrow_forwardGive the product or products that you would expect from the following reaction. In each case give the mechanism as SN1, SN2, E1, E2 and predict the relative amount of each product. - (CH3)3CCl+OH- / CH3OH -->arrow_forwardWhat is the predominant mechanism of the reaction below? SN2 SN1 E2 E1arrow_forward
