
Interpretation:
The reason has to be explained for formation of racemic mixture of 2-bromooctane when the reaction of NaBr with (R)-2-Bromooctane.
Concept introduction:
SN2 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid, the bromine atom attacks back side of the carbon atoms in simultaneous manner and which is bearing alcohol group which yield the corresponding product.
Example:
The tosylated compound is reaction with sodium methoxide which undergoes again type of reaction, the methoxide ion attacks the carbon atom through the back side and provides Inverse configuration of methoxy compound. This is shown below,
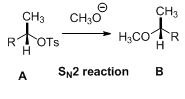
Given information:
The given compound is shown below,
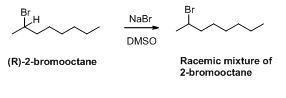
Trending nowThis is a popular solution!

Chapter 11 Solutions
Organic Chemistry
- iii) 2-Bromo-2-cyclopropylpropane will undergo an SN1 reaction called solvolysis in methanol to give several products, two of which are shown below. Use curly arrows to show how the formation of these two products occurs mechanistically.arrow_forward1-Bromobutane would react with HS- via _______ reactiona.) SN1 b.) SN2 c.) E1 d.) E2arrow_forwardPropose a plausible mechanism for the 1 to 2 conversion as shown, and explain the stereochemical result:arrow_forward
- Optically active butan-2-ol racemizes in dilute acid. Propose a mechanism for this racemization.arrow_forwardWhat are the products obtained (2 stereoisomers A and B) when (Z)-hex-3-ene is treated with Br2 in anhydrous medium? Show the mechanism of this reaction. When the dibromide formed is treated with KOH in EtOH, only (Z)-3-bromohex-3-ene. Explain this result and justify why the E isomer cannot be obtained here. here.arrow_forwardThe reaction of (S)-2-bromopentane with potassium cyanide to yield 2-methylpentanenitrile (2-cyanopentane) occurs via a nucleophilic substitution pathway. The reaction is 100% stereospecific. Please explain in complete words what this observation tells you about the mechanism of the reaction.arrow_forward
- Identify the products obtained when compound 1 is hydrolyzed in the presence of an acid and provide a detailed reaction mechanism for this hydrolysis of compound 1.arrow_forwardWhich one of the following compounds gives 5-methyl-3-heptanol with LiAlH4 followed by water?arrow_forwardHow many distinct alkene products are possible when the alkyl iodide below undergoes E2 elimination?arrow_forward
- When 5-bromo-1-pentanol is treated with sodium hydride in diethyl ether, the product is analyzed to be C5H10O. Propose a likely structure for this product, suggesting a reasonable mechanistic pathway for its formationarrow_forwardExplain the following result. Although alkenes are generally more reactive than alkynes towards electrophiles, the reaction of Cl2 with but-2-yne can be stopped after one equivalent of Cl2 has been added.arrow_forwardWhat is the major product obtained from treating an excess of each of the following compounds with Cl2 in the presence of ultraviolet light at roomtemperature? Disregard stereoisomers.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

