
a)
Interpretation:
The actual product has to be identified.
Concept introduction:
Elimination reaction:An elimination reaction is removal of two substituents in a molecule and forms
E1 elimination:
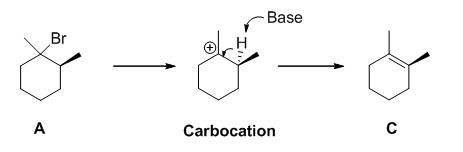
E2 elimination:Alkyl halide forms an alkene from the abstraction of the proton from the β-carbon atom followed by elimination of the bromine in a single step.
b)
Interpretation:
The actual product has to be identified.
Concept introduction:
Elimination reaction:An elimination reaction is removal of two substituents in a molecule and forms alkene. An elimination reaction is one or two-step process which based on the mechanism when two substituents removed from the molecule in single step is called E2 reaction. When two substituents are removed from the molecule in two steps is called E1 reaction.
E1 elimination: Alkyl halide forms carbocation by the removal of bromine followed by the abstraction of the proton from the β-carbon atom in two steps which leads to the product as an alkene

E2 elimination:Alkyl halide forms an alkene from the abstraction of the proton from the β-carbon atom followed by elimination of the bromine in a single step.

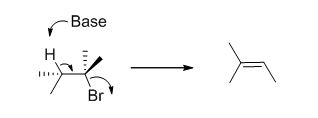
c)
Interpretation:
The actual product has to be identified.
Concept introduction:
Elimination reaction:An elimination reaction is removal of two substituents in a molecule and forms alkene. An elimination reaction is one or two-step process which based on the mechanism when two substituents removed from the molecule in single step is called E2 reaction. When two substituents are removed from the molecule in two steps is called E1 reaction.
E1 elimination: Alkyl halide forms carbocation by the removal of bromine followed by the abstraction of the proton from the β-carbon atom in two steps which leads to the product as an alkene

E2 elimination:Alkyl halide forms an alkene from the abstraction of the proton from the β-carbon atom followed by elimination of the bromine in a single step.

Trending nowThis is a popular solution!

Chapter 11 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- give the products and reasonable mechanisms for the following reactions. What are the names of these two reactions?arrow_forwardCan you help predict the product from these reactions?arrow_forwardC(CH3)3X +H20 where (X-F, CI, Br, I) give reaction with each of halogen as well determine with which halogen substitution reaction is more favorable and feasible and give reason of its feasibility? C(CH3)3OHarrow_forward
- Consider a hypothetical chemical reaction between compound A and compound B, which produces compound C as the final product. The reaction is known to be exothermic and spontaneous. However, when the reaction is carried out under certain conditions, it fails to occur. Explain this observation and propose a potential solution to overcome this hurdle.arrow_forward5b. Enolates have two resonance structures, and can react on oxygen instead of carbon. Show the product of reaction in the oxygen, and then show how it can be converted to the product with the allyl group on carbon.arrow_forward3. cyclohexanone to cyclohexane. A second year chemistry student is assigned a task to prepare compound 2 from compound 1 using lithium aluminium hydride as a reducing agent. However, from the reaction mixture, no traces of product 2 were detected. Explain this observation in detail and suggest an alternative route that will lead to product 2. H 1 CH3 LiAllH4 H O: 2 CH3 quearrow_forward
- Predict one or two main products from each reaction belowarrow_forwardTreatment of (HOCH,CH,CH,CH,),CO with acid forms a product of molecular formula C3H1,02 and a molecule of water. Draw the structure of the product and explain how it is formed.arrow_forwardPredict the product of the following reactions: Note also include the by-products.arrow_forward
- Each of the following compounds will undergo nucleophilic substitution, replacing the Cl with a methoxide (OCH) group. Predict which compound A, B, Cor D will react the fastest and which compound will react the slowest under the reaction conditions listed below (there is no need to rank all 4). Explain your reasoning and provide structures and intermediates to support your conclusion. CH,OH nucelophilic substitution heat Darrow_forwardPredict the products and include stereochemistry.arrow_forwardPredict the product of the reaction. Include all hydrogen atomsarrow_forward
