
a)
Interpretation:
The given molecule has to be prepared by using nucleophilic substitution reaction.
Concept introduction:
SN1 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid which yield the corresponding carbocation intermediate, this carbocation intermediate undergoes substitution reaction which yields the corresponding substitution product.
Tertiary alcohols undergo substitution very fast than the secondary alcohols because tertiary carbocation is more stable than the secondary carbocation than the primary carbocation.
Primary alcohol is less stable therefore it won’t undergo SN1substitution reaction.
SN2 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid, the bromine atom attacks back side of the carbon atoms in simultaneous manner and which is bearing alcohol group which yield the corresponding product.
Example:
Alcohol is reaction with tosyl chloride in pyridine which provides retention of configuration of tosylated compound. This tosylated compound is further reaction with sodium methoxide which undergoes again SN2 type of reaction, the methoxide ion attacks the carbon atom through the back side and provides Inverse configuration of methoxy compound. This is shown below,
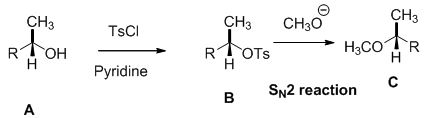
SN2 reaction is second order
Answer to Problem 45AP
The reaction is given below,
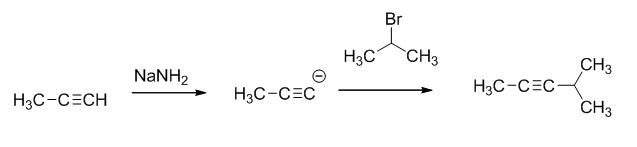
Explanation of Solution
Given information:
The product of the reaction is given below,
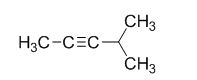
The reaction is given below,
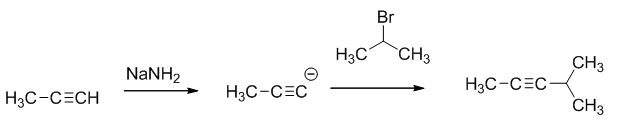
The sodium amide is acts as a base and it abstract the highly acidic proton from propylene gives carbanion, this carbanion react with 2-bromo propane gives the corresponding product.
The given molecule is prepared by using nucleophilic substitution reaction.
b)
Interpretation:
The given molecule has to be prepared by using nucleophilic substitution reaction.
Concept introduction:
SN1 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid which yield the corresponding carbocation intermediate, this carbocation intermediate undergoes substitution reaction which yields the corresponding substitution product.
Tertiary alcohols undergo substitution very fast than the secondary alcohols because tertiary carbocation is more stable than the secondary carbocation than the primary carbocation.
Primary alcohol is less stable therefore it won’t undergo SN1substitution reaction.
SN2 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid, the bromine atom attacks back side of the carbon atoms in simultaneous manner and which is bearing alcohol group which yield the corresponding product.
Example:
Alcohol is reaction with tosyl chloride in pyridine which provides retention of configuration of tosylated compound. This tosylated compound is further reaction with sodium methoxide which undergoes again SN2 type of reaction, the methoxide ion attacks the carbon atom through the back side and provides Inverse configuration of methoxy compound. This is shown below,

SN2 reaction is second order reaction, the rate of the reaction is depending on the both substrate and nucleophiles.
Answer to Problem 45AP
The reaction is given below,
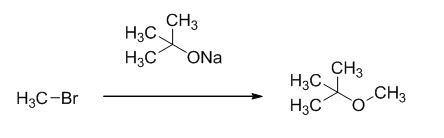
Explanation of Solution
Given information:
The product of the reaction is given below,
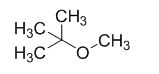
The reaction is given below,
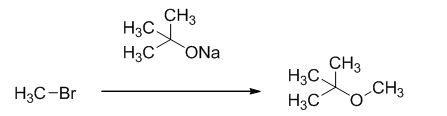
The sodium tertiary butoxide is acts as a base and it react with methyl bromide gives the corresponding ether product.
The given molecule is prepared by using nucleophilic substitution reaction.
c)
Interpretation:
The given molecule has to be prepared by using nucleophilic substitution reaction.
Concept introduction:
SN1 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid which yield the corresponding carbocation intermediate, this carbocation intermediate undergoes substitution reaction which yields the corresponding substitution product.
Tertiary alcohols undergo substitution very fast than the secondary alcohols because tertiary carbocation is more stable than the secondary carbocation than the primary carbocation.
Primary alcohol is less stable therefore it won’t undergo SN1substitution reaction.
SN2 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid, the bromine atom attacks back side of the carbon atoms in simultaneous manner and which is bearing alcohol group which yield the corresponding product.
Example:
Alcohol is reaction with tosyl chloride in pyridine which provides retention of configuration of tosylated compound. This tosylated compound is further reaction with sodium methoxide which undergoes again SN2 type of reaction, the methoxide ion attacks the carbon atom through the back side and provides Inverse configuration of methoxy compound. This is shown below,

SN2 reaction is second order reaction, the rate of the reaction is depending on the both substrate and nucleophiles.
Answer to Problem 45AP
The reaction is given below,
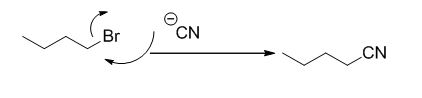
Explanation of Solution
Given information:
The product of the reaction is given below,
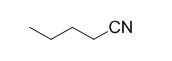
The reaction is given below,
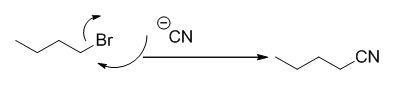
n-butyl bromide undergoes nucleophilic substitution reaction with cyanide (sodium cyanide) gives the corresponding cyanide product.
The given molecule is prepared by using nucleophilic substitution reaction.
d)
Interpretation:
The given molecule has to be prepared by using nucleophilic substitution reaction.
Concept introduction:
SN1 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid which yield the corresponding carbocation intermediate, this carbocation intermediate undergoes substitution reaction which yields the corresponding substitution product.
Tertiary alcohols undergo substitution very fast than the secondary alcohols because tertiary carbocation is more stable than the secondary carbocation than the primary carbocation.
Primary alcohol is less stable therefore it won’t undergo SN1substitution reaction.
SN2 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid, the bromine atom attacks back side of the carbon atoms in simultaneous manner and which is bearing alcohol group which yield the corresponding product.
Example:
Alcohol is reaction with tosyl chloride in pyridine which provides retention of configuration of tosylated compound. This tosylated compound is further reaction with sodium methoxide which undergoes again SN2 type of reaction, the methoxide ion attacks the carbon atom through the back side and provides Inverse configuration of methoxy compound. This is shown below,

SN2 reaction is second order reaction, the rate of the reaction is depending on the both substrate and nucleophiles.
Answer to Problem 45AP
The reaction is given below,
Explanation of Solution
Given information:
The reaction is given below,
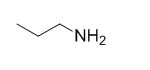
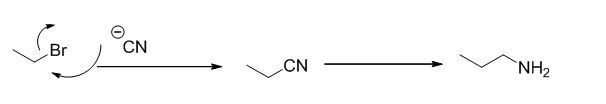
The reaction is given below,
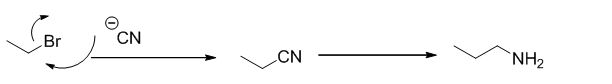
Ethyl bromide undergoes nucleophilic substitution reaction with cyanide (sodium cyanide) gives the corresponding cyanide product, this cyanide undergoes catalytic reduction using Pt (metal reduction) gives
The given molecule is prepared by using nucleophilic substitution reaction.
Want to see more full solutions like this?
Chapter 11 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- Consider the following groups of compounds. In each series circke the weaker (poorer) nucleophile. Please explain why.arrow_forwardExplain why each of the following reactions will not form the given product. Then, design a synthesis of A from benzene and B from phenol (C6H5OH).arrow_forwardIdentify the electrophile and the nucleophile in each of the following reaction steps. Then draw curved arrows to illustrate the bond-making and bond-breaking processes.arrow_forward
- Which of these compounds would undergo nucleophilic aromatic substitution the fastest?arrow_forwardSynthesize the resulting product using the following compounds using the appropriate reaction steps.arrow_forwardRank the following species in each set from best nucleophile to poorest nucleophile.arrow_forward
- What is the slow (rate-determining) step in any electrophilic aromatic substitution reaction? Please provide a detailed explanation.arrow_forwardFrom compounds of 4 carbons or less and any reagents you want; synthesize the following compound.arrow_forwardWhen a single compound contains both a nucleophile and a leaving group, an intramolecular reaction may occur. With this in mind, draw the product of the following reactionarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
