
Interpretation:
The structure of the isomer has to be drawn and named for the given compound and also decreasing order of reactivity in the SN2 reaction has to arranged.
Concept introduction:
Isomer:
A molecule having same molecular formula with different chemical formula is called isomer.
Constitutional Isomers:
A molecule having same molecular formula with different structural formulas (Difference in the connectivity of the molecule is called constitutional isomer).
SN2 reaction:
Tosylated compound is reaction with sodium methoxide which undergoes again type of reaction, the methoxide ion attacks the carbon atom through the back side and provides Inverse configuration of methoxy compound. This is shown below,
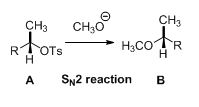
Given information:
The given isomers of molecular formula of C4H9Br.
Trending nowThis is a popular solution!

Chapter 11 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- Make a table with the main conditions for each mechanism: E1, E2, SN1, SN2.arrow_forward2) Rank the following compounds in order of their reaction rates in an SN1 reaction with NaF with 1 being the fastest reaction and 5 being the slowest. OTs Br CIarrow_forwardOrder the compounds from most to least reactive in SN2 reactions. Explain why.arrow_forward
- Which of the following compounds will undergo the fastest SN1 reaction? A B IV || C) III E Both I & IV || Br E IV Brarrow_forwardNot sure what substitution goes here for the SN2 reactionarrow_forwardA student adds NBS to a solution of 1-methylcyclohexene and irradiates the mixture with a sunlamp until all the NBS has reacted. After a careful distillation, the product mixture contains two major products of formula C7H11Br. (a) Draw the resonance forms of the three possible allylic free radical intermediates.arrow_forward
- 9. Which SN1 reaction in each pair is faster? a) CI b) (CH3)3CCI + H2O > H2O (CH3)3CBR + H20 → CI H,O 206arrow_forwardrank the following compounds in order of their expected reactivity toward SN2 reaction CH3Br CH3OTos (CH3)3CCl (CH3)2CHClarrow_forwardList the following in increasing order of nucleophilic strength. CH3COO-, H2O, CH3O-, OH-, CH3CH20-arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





