
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
thumb_up100%
Chapter 11.SE, Problem 62AP
Interpretation Introduction
Interpretation:
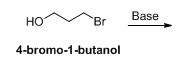
The product has to be identified when 4-bromo-1-butanol reacts with base.
Concept introduction:
SN2 reaction:
The alcohols is reaction with acids like hydrochloric acid or hydrobromic acid, the bromine atom attacks back side of the carbon atoms in simultaneous manner and which is bearing alcohol group which yield the corresponding product.
Example:
The tosylated compound is reaction with sodium methoxide which undergoes again SN2 type of reaction, the methoxide ion attacks the carbon atom through the back side and provides Inverse configuration of methoxy compound. This is shown below,
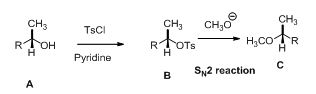
Given information:
The given compound is shown below,
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What is the products you expect from oxidation of 2-Hexanol with CrO3 and H3O+?
What reaction conditions are needed to convert (R)-2-ethyl-2- methyloxirane to (R)-2-methylbutane-1,2-diol ?
How would you carry out the following reactions? More than one step may be required.
Benzene -> m-bromoacetophenone
1-methylcyclohexene -> 2-methylcyclohexanone
Chapter 11 Solutions
Organic Chemistry
Ch. 11.1 - Prob. 1PCh. 11.2 - Prob. 2PCh. 11.2 - Prob. 3PCh. 11.3 - Prob. 4PCh. 11.3 - Prob. 5PCh. 11.3 - Rank the following compounds in order of their...Ch. 11.3 - Organic solvents like benzene, ether, and...Ch. 11.4 - Prob. 8PCh. 11.4 - Prob. 9PCh. 11.4 - Prob. 10P
Ch. 11.5 - Rank the following substances in order of their...Ch. 11.5 - 3-Bromo-1-butene and 1-bromo-2-butene undergo SN1...Ch. 11.5 - Prob. 13PCh. 11.6 - Review the mechanism of geraniol biosynthesis...Ch. 11.7 - Prob. 15PCh. 11.7 - What alkyl halides might the following alkenes...Ch. 11.8 - Prob. 17PCh. 11.8 - Prob. 18PCh. 11.9 - Prob. 19PCh. 11.12 - Prob. 20PCh. 11.SE - Prob. 21VCCh. 11.SE - From what alkyl bromide was the following alkyl...Ch. 11.SE - Prob. 23VCCh. 11.SE - Prob. 24VCCh. 11.SE - Prob. 25MPCh. 11.SE - Prob. 26MPCh. 11.SE - Prob. 27MPCh. 11.SE - Prob. 28MPCh. 11.SE - Prob. 29MPCh. 11.SE - Prob. 30MPCh. 11.SE - Prob. 31MPCh. 11.SE - Prob. 32MPCh. 11.SE - Metabolism of S-adenosylhomocysteine (Section...Ch. 11.SE - Reaction of iodoethane with CN- yields a small...Ch. 11.SE - One step in the urea cycle for ridding the body of...Ch. 11.SE - Prob. 36MPCh. 11.SE - Prob. 37MPCh. 11.SE - Propose a mechanism for the following reaction, an...Ch. 11.SE - Prob. 39APCh. 11.SE - The following Walden cycle has been carried out....Ch. 11.SE - Prob. 41APCh. 11.SE - Which reactant in each of the following pairs is...Ch. 11.SE - Prob. 43APCh. 11.SE - Prob. 44APCh. 11.SE - Prob. 45APCh. 11.SE - Prob. 46APCh. 11.SE - Prob. 47APCh. 11.SE - Prob. 48APCh. 11.SE - Propose structures for compounds that fit the...Ch. 11.SE - What products would you expect from the reaction...Ch. 11.SE - Prob. 51APCh. 11.SE - Prob. 52APCh. 11.SE - Prob. 53APCh. 11.SE - Prob. 54APCh. 11.SE - Prob. 55APCh. 11.SE - Order each of the following sets of compounds with...Ch. 11.SE - Order each of the following sets of compounds with...Ch. 11.SE - Prob. 58APCh. 11.SE - Prob. 59APCh. 11.SE - Ethers can often be prepared by SN2 reaction of...Ch. 11.SE - Show the stereochemistry of the epoxide (see...Ch. 11.SE - Prob. 62APCh. 11.SE - In addition to not undergoing substitution...Ch. 11.SE - The tosylate of (2R, 3S)-3-phenyl-2-butanol...Ch. 11.SE - Prob. 65APCh. 11.SE - Prob. 66APCh. 11.SE - Prob. 67APCh. 11.SE - Prob. 68APCh. 11.SE - Prob. 69APCh. 11.SE - (S)-2-Butanol slowly racemizes on standing in...Ch. 11.SE - Reaction of HBr with (R)-3-methyl-3-hexanol leads...Ch. 11.SE - Treatment of 1-bromo-2-deuterio-2-phenylethane...Ch. 11.SE - Prob. 73APCh. 11.SE - Prob. 74APCh. 11.SE - In light of your answer to Problem 11-74, explain...Ch. 11.SE - Prob. 76APCh. 11.SE - Compound X is optically inactive and has the...Ch. 11.SE - When a primary alcohol is treated with...Ch. 11.SE - Prob. 79APCh. 11.SE - Amines are converted into alkenes by a two-step...Ch. 11.SE - The antipsychotic drug flupentixol is prepared by...
Knowledge Booster
Similar questions
- Show how to convert (E)-hex-3-ene into propyl propionate. You must use (E)-hex-3-ene as the source of all carbon atoms in the target molecule. Show all reagents and all molecules synthesized along the way.arrow_forwardChoose a Grignard reagent _______and a ketone _______ that can be used to produce the following compound: 1-Ethylcyclohexanolarrow_forwardwhat products would be expected if (R)-2-butanol were heated with concenrated hydrobromic acid? if more than one product is expected what will be the major product?arrow_forward
- How can you prepare 2-methyl-2-butanol starting with 3-methyl-1-butyne?arrow_forwardReaction of 2-methyl-1-hexene with 1) m-Chloroperbenzoic acid followed by 2) addition of hydronium ion will provide Group of answer choices a) an enantiomeric mixture of 2-methyl-1,2-hexanediol b) an enantiomeric mixture of 2-methyl-2,3-hexanediol c) 2-methylhexan-2-ol d) 2-methyl-1,2-hexanediol e) an enantiomeric mixture of 2-methylhexan-2-olarrow_forwardAnswer these two questions with explanation: (10 pts) 1. When an aldehyde or ketone is treated with diol such as ethylene glycol (1,2-ethanediol) and an acid catalyst, a cyclic acetal is formed. Draw the structure of the product you would obtain from benzaldehyde and ethylene glycol. 2. Would you expect hemiacetals to be stable in basic solutions? Explain your answer (Hint: examine the mechanism of formation of hemiacetals)arrow_forward
- Which of the following synthetic routes will convert 3-methyl-2-butanol into 3-methyl-1-butanol?arrow_forwardWhich of the reagents would accomplish the transformation of 2-methyl-3-cyclopentanol into 2-methyl-3-cyclopentenone? A.) Br2, CCl4 B.) KMnO4 C.) H2, Pt D.) CrO3/H2SO4arrow_forwardIn addition to not undergoing substitution reactions, the alkyl bromide shown in Problem 11-32 also fails to undergo an elimination reaction when treated with base. Explain.arrow_forward
- How would you carry out the following reactions? More than one step may be required. (a) 3-Hexyne → 3-Hexanone (b) Benzene → m-Bromoacetophenone (c) Bromobenzene → Acetophenone (d) l-Methylcyclohexene → 2-Methylcyclohexanonearrow_forwardWhat products would you expect from the reaction of 1-bromopropane with each of the following? (a) NaNH2 (b) KOC(CH3)3 (c) NaI (d) NaCN (e) NaCCH (f) Mg, then H2Oarrow_forwardUsing your reaction roadmap as a guide, show how to convert 3-hexyne into propanal. All of the carbon atoms of the target molecule must be derived from the starting material as efficiently as possible. Show all intermediate molecules synthesized along the way.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning