
(a)
Interpretation:
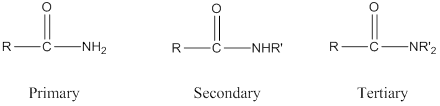
The structure of an amide with formula
Concept Introduction:
Amide: One
Depending on the number of carbon side chain of the amide, different types of amides can form.
(b)
Interpretation:
The structure of an
Concept Introduction:
Functional group: They are certain substitutes in the organic molecules which are determine the characteristic reactions taking place in it.
Aldehyde: One alkyl group and a hydrogen atom are attached to the carbonyl carbon atom of the compound. It is represented as
(c)
Interpretation:
The structure of an aromatic compound which is an ether with molecular formula
Concept Introduction:
Functional group: They are certain substitutes in the organic molecules which are determine the characteristic reactions taking place in it.
Ether: Ether is a group of organic compound where two aryl or alkyl groups are connected by an oxygen atom. It is represented as
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
EBK FUNDAMENTALS OF GENERAL, ORGANIC, A
- Draw structural formulas for all of the following. Q.) Alcohols with the molecular formula C4H10Oarrow_forwardDraw condensed structural formulas for the two carboxylic acids with the molecular formula C4H8O2arrow_forwardCompound A undergoes a reaction with hydrogen bromide, HBr to produce2-bromobutane. A exists as cis-trans isomers and decolourises brominesolution in methylene chloride, CH2Cl2. a)Draw and name the structure of compound D. b)Draw two (2) constitutional isomers of compound Darrow_forward
- Identify the functional groups in the following molecules. (Use names from the table below. List each class of functional group only once. If there are fewer than 3 functional groups, leave an appropriate number of answer boxes empty.) а) NH2 phenylalanine CH3 .CH3 b) ÕH ephedrinearrow_forwardCompute the formal charge (FC) on each atom in the following structures [H2CNH2]+arrow_forwardWrite the possible structural formulas for all the constitutionally isomeric compounds having the given molecular formula USING THE DASH FORMULA. Take into account the valency of each atom. Indicate the Formal Charge of the Atom, if there is any on the proposed lewis structure. 2. C5H12O (at least 1 ether, at least 1 Alcohol)arrow_forward
- Use Frost Circles to complete the molecular orbital diagram for cyclooctatetrane. Label the bonding, non bonding, and anti bonding MO’s. If the molecule is planar, would it be aromatic, antiaromatic, or nonaromatic? If the molecule is nonplanar, would it be aromatic, antiaromatic, or nonaromatic?arrow_forwardHow many potential stereoisomers exist for the following molecule? HQ A) 3 B) 6 C) 8 D) 11 O A O B OC ODarrow_forwardGive structures for the following, based on the data in Table . (a) cis-9-Dodecenoic acid (b) 18:1c∆11 (c) A saturated fatty acid that should melt below 30 °Carrow_forward
- Define the following terms:(a) Chiral (b) Achiral(c) Chiral carbon (d) Enantiomerarrow_forwardSketch the MO diagram for 1,3 budadiene. Determine the net bonding interactions for ¥2 and ¥3. How many nodes are in ¥4? How many AO’s were used? How many MO’s resulted?arrow_forwardHow many peptide bonds were produced by creating the following How many H2O particle were produced by creating the following structure if the starting reagent is glucose? * structure if the starting reagent is glucose? * CH,OH CH,OH CH,OH CH;OH CH;OH CH;OH OH он он OH 250 ÓH OH OH ÓH OH OH 300 А.) 125 B.) 250 А.) 30 c.) 25 B.) 301 D.) 500 C.) 302 E.) O D.) 300 E.) 150 F.) Oarrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





