
(a)
Interpretation:
The structure of an amide with formula
Concept Introduction:
Functional group: They are certain substitutes in the organic molecules which are determine the characteristic reactions taking place in it.
Amide: One
Depending on the number of carbon side chain of the amide, different types of amides can form.
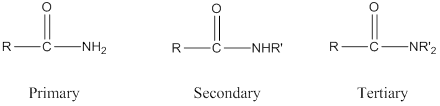
(b)
Interpretation:
The structure of an
Concept Introduction:
Functional group: They are certain substitutes in the organic molecules which are determine the characteristic reactions taking place in it.
Aldehyde: One alkyl group and a hydrogen atom are attached to the carbonyl carbon atom of the compound. It is represented as
(c)
Interpretation:
The structure of an aromatic compound which is an ether with molecular formula
Concept Introduction:
Functional group: They are certain substitutes in the organic molecules which are determine the characteristic reactions taking place in it.
Ether: Ether is a group of organic compound where two aryl or alkyl groups are connected by an oxygen atom. It is represented as
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
- Give the molecular formula of the functional group that is missing. a. NH+ b. CH3 c. COOH d. C6H12O6arrow_forwardProvide compounds that fit the following descriptions:(a) Two amines that are gases at room temperature(b) A heterocyclic amine(c) A compound with an amine group on an aromatic ringarrow_forwardWhen Alfred Werner was developing the field of coordination chemistry, it was argued by some that the optical activity he observed in the chiral complexes he had prepared was due to the presence of carbon atoms in themolecule. To disprove this argument, Werner synthesized a chiral complex of cobalt that had no carbon atoms in it, and he was able to resolve it into its enantiomers. Design a cobalt(III) complex that would be chiral if it could be synthesized and that contains no carbon atoms.arrow_forward
- Write the possible structural formulas for all the constitutionally isomeric compounds having the given molecular formula USING THE DASH FORMULA. Take into account the valency of each atom. Indicate the Formal Charge of the Atom, if there is any on the proposed lewis structure. 2. C5H12O (at least 1 ether, at least 1 Alcohol)arrow_forwardGive IUPAC names for the five isomers with the formula C6H14.arrow_forwardWrite the structure of L-Glutamic acid in pH 9 Write the Structure of L-Lysine in pH 1 2. Similarly write equations to show how L-lysine and L-Glutamic acid gives products when reacted with OH- ion as well as H3O+ ionarrow_forward
- Draw the complete structural formula of arachidonic acid (Table 23.1) in a way that shows the cis stereochemistry of its four double bonds.arrow_forwardEthylene glycol, the main ingredient in antifreeze, contains 38.7% carbon, 9.7% hydrogen and 51.6 % oxygen. Calculate the empirical and molecular formulas for ethylene glycol. Given the molar mass is approximately 60 g/mol. A) Empirical formula: B)Molecular formula: Explain how you obtained the Molecular formula (b)?arrow_forwardButadiene (right) is a colorless gas used to make synthetic rubber and many other compounds. (a) How many σ bonds and π bonds does the molecule have? (b) Are cis-trans arrangements about the double bonds possible? Explain.arrow_forward
- Drawn are four isomeric dimethylcyclopropanes. How are the compounds in each pair related (enantiomers, diastereomers, constitutional isomers): A and B; A and C; B and C; C and D?arrow_forwardCompute the formal charge (FC) on each atom in the following structures [H2CNH2]+arrow_forwardConvert the following structural formulas into condensed structures.arrow_forward
 Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning
Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning


