
Interpretation:
The mechanism for the reaction in Equation 12-17 is as follows, but the curved arrows have been omitted. The mechanism by drawing in the curved arrows is to be completed. Also, the name of each elementary step below the appropriate reaction arrow is to be written.
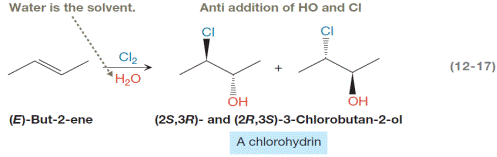
Concept introduction:
A chlorohydrin is produced when an
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
ORGANIC CHEM. PRIN AND MECHANISM LL+ AC
- Draw the mechanism for this reactionarrow_forwardConsider the following reactants: X Would elimination take place at a significant rate between these reactants? oyes X 5 no If you said elimination would take place, draw the major products in the upper drawing area. If you said elimination would take place, also draw the complete mechanism for one of the major products in the lower drawing area. If there is more than one major product, you may draw the mechanism that leads to any of them. Major Products: Mechanism: Click and drag to start drawing a structure. Add/Remove step 0: Click and drag to start drawing a structure. D + םarrow_forwardPlease help me with a drawn mechanism for the shown reaction.arrow_forward
- 6. Consider the following E1 reaction. OH H3O+ reflux + H₂O a. Draw the complete, detailed mechanism (curved arrows) for this reaction. b. Draw a reaction free energy diagram that agrees with the mechanism, labeling overall reactants, overall products, all transition states, and all intermediates.arrow_forwardDraw a complete, detailed mechanism for the following reaction. A key intermediate is provided.arrow_forwardPredict the relative rates of these reactions. That is, select 1 next to the reaction with the fastest rate, 2 next to the reaction with the next fastest rate, and so on. Note for advanced students: you may assume these reactions all take place in a polar aprotic solvent, like DMSO. ( Choose one) Predict the relative rates of these reactions. That is, select 1 next to the reaction with the fastest rate, 2 next to the reaction with the next fastest rate, and so on. Note for advanced students: you may assume these reactions all take place in a polar aprotic solvent, like DMSO. u + H₂O Reaction - OH₂ Relative Rate (Choose one) OH₂ (Choose one) ▼ +CI ... + H₂O OH + OH + I (Choose one) (Choose one) ▼ + H₂S + CIarrow_forward
- The reaction shown here proceeds via a carbocation rearrangement. Draw a complete, detailed mechanism to account for the product. Explain why the carbocation rearrangement is favorable. CH;OH Brarrow_forwardShown below is a two-step mechanism beginning with nucleophilic attack of water, and subsequent deprotonation with a base. Draw the arrows for the mechanism for both step 1 and step 2 and draw the intermediate product of in the box. HH H-O OH H₂O Barrow_forwardgiven reaction (see image), draw the curly arrows for the mechanism to show the movement of electron pairsarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
