
- 13. It is difficult to bring the Internet to some remote parts of the world. This can be inexpensively done by installing antennas tethered to large helium balloons. To help analyze the situation, assume we have inflated a large spherical balloon. The pressure on the inside of the balloon is balanced by the elastic force exerted by the rubberized material. Since we are dealing with a gas in an enclosed space, the ideal gas law will be applicable.
PV=nRT
Where
P = pressure [atm]
V = volume [L]
n = quant ity of gas [moles]
R = ideal gas constant [0.08206 (atm L)/ (mol K)]
T = temperature [K]
If the temperature increases, the balloon will expand and/or the pressure will increase to maintain the equality. As it turns out, the increase in volume is the dominant effect, so we will treat the change in pressure as negligible.
The circumference of an inflated spherical balloon is measured at various temperatures; the resulting data are shown in the following graph.
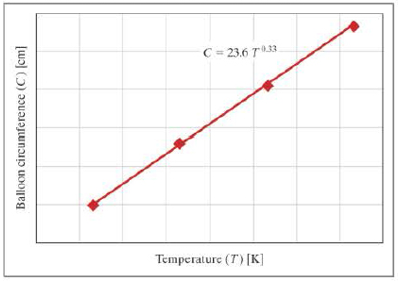
- a. What are the units of the constant 0.33?
- b. What are the units of the constant 23.6?
- c. What would the temperature of the balloon be if the circumference was 162 centimeters?
- d. If a circle with an area of 100 square centimeters is drawn on the balloon at 20 degrees Celsius, what would the area be at a temperature of 100 degrees Celsius?
- e. If the pressure inside the balloon is 1.2 atmospheres, how many moles of gas does it contain?
Trending nowThis is a popular solution!

Chapter 12 Solutions
Thinking Like an Engineer: An Active Learning Approach (4th Edition)
Additional Engineering Textbook Solutions
Fluid Mechanics Fundamentals And Applications
Shigley's Mechanical Engineering Design (McGraw-Hill Series in Mechanical Engineering)
Automotive Technology: Principles, Diagnosis, and Service (5th Edition)
Mechanics of Materials, 7th Edition
Fundamentals Of Thermodynamics
Introduction To Finite Element Analysis And Design
- The atmospheric pressure on the surface of Mars is only 0.007 bar which is less than 1% of the Earth’s atmospheric pressure. What is the main gas in the atmosphere of Mars. A. Carbon Dioxide B. Hydrogen C. Helium D. Nitrogen E. Sulfur Dioxidearrow_forwardAccording to Newton's 3rd Law, an action and reaction are equal in____. weight shape sizearrow_forwardWhich of the following statement(s) is(are) correct? Group of answer choices A. N B. The pressure, volume, and temperature of the steam are not connected by simple equations unlike ideal gases. C. An irreversible adiabatic process is a constant entropy process. D. The vaporization process of steam is isobaric in nature. E. The general energy equations for ideal gas are analogous to the equations for the processes of vapors.arrow_forward
- 20.) When a process proceeds in such a manner that the system remains infinitesimally close to an equilibrium state at all times, it is called quasi-equilibrium process. Select one: True Falsearrow_forwardquestion 1) 1. Describe how the mean effective pressure for reciprocating engines is defined and its importance for thermodynamic analysis of internal combustion engines.arrow_forward5. What is the degree of freedom if the number of independent equations are 5 and the number of unknowns is 7. Is the problem over-specified or under-specified? 6. Write a general statement for the unsteady state mass balance. Write a similar statement describing activity in a bank account related to monetary balance.arrow_forward
- 3 A farmer has a 10-heactare maize farm. The maize is under “Fall Army Worm” attack. The farmer has to urgently control the attack using a chemical called AGENOX 221. Motorized sprayers with tank capacities of 15 litres each are to be used. The recommended concentration of the chemical is 2 ml/L and each full tank will cover ½ hectare. A 50-man team is engaged to control the attack. Using the above information and the assumptions given below: i. What volume of the chemical is required to cover the entire field to control the “Fall Army Worm” attack? ii. If each man covers an area of 25 m2 from one end of the field to the other, how many trips will each of them make to cover the entire field? Assume ach man moves in a straight line. Assumptions: 1. The 10 ha farm is considered to be a perfect square. 2. There is a 2.5 % loss of chemical due to the transfer to the sprayer tank.arrow_forwardselect the incorrect statement a. work and heat transfer represent energy crossing a boundary. b. differential of work and heat transfer are exact. c. work and heat transfer are path integral. d. net work and net heat transfer are equal for a cycle.arrow_forwardStefan-Boltzmann law. Thermal radiation by a grey body.arrow_forward
 Principles of Heat Transfer (Activate Learning wi...Mechanical EngineeringISBN:9781305387102Author:Kreith, Frank; Manglik, Raj M.Publisher:Cengage Learning
Principles of Heat Transfer (Activate Learning wi...Mechanical EngineeringISBN:9781305387102Author:Kreith, Frank; Manglik, Raj M.Publisher:Cengage Learning Refrigeration and Air Conditioning Technology (Mi...Mechanical EngineeringISBN:9781305578296Author:John Tomczyk, Eugene Silberstein, Bill Whitman, Bill JohnsonPublisher:Cengage Learning
Refrigeration and Air Conditioning Technology (Mi...Mechanical EngineeringISBN:9781305578296Author:John Tomczyk, Eugene Silberstein, Bill Whitman, Bill JohnsonPublisher:Cengage Learning

