
Concept explainers
Eutrophication is a process whereby lakes, estuaries, or slow-moving streams receive excess nutrients that stimulate excessive plant growth. This enhanced plant growth, often called an algal bloom, reduces dissolved oxygen in the water when dead plant material decomposes and can cause other organisms to die. Nutrients can come from many sources, such as fertilizers; deposition of nitrogen from the atmosphere; erosion of soil containing nutrients; and sewage treatment plant discharges. Water with a low concentration of dissolved oxygen is called hypoxic. A biosystems engineering models the algae growth in a lake. The concentration of algae (C), measured in grams per milliliter (g/ml], can be calculated by
Where
Co = initial concentration of algae [?]
K= multiplication rate of the algae [?]
r= estimated nutrient supply amount [mg of nutrient per mL of sample water]
t= time [days]
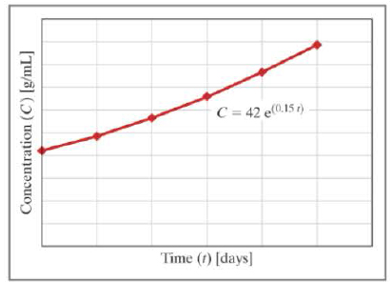
- a. For the exponential model shown, list the value and units of the parameters m and b . You do not need to simplify any units. Recall that an exponential model has the form: y=bemx .
- b. What are the units on the multiplication rate of the algae (k)?
- c. If the algae are allowed to grow for 10 days with an estimated nutrient supply of 3 milligrams of nutrient per milliliter of water sample, what is the multiplication rate of the algae (k)?
Trending nowThis is a popular solution!

Chapter 12 Solutions
Thinking Like an Engineer: An Active Learning Approach (4th Edition)
Additional Engineering Textbook Solutions
Foundations of Materials Science and Engineering
Mechanics of Materials, 7th Edition
Mechanics of Materials
Applied Fluid Mechanics (7th Edition)
Manufacturing Engineering & Technology
Thermodynamics: An Engineering Approach
- What does it mean for an engineering device that has steady-flow? (choose 3 answers) 1- it has been operating for a long time. 2- the properties do not change with time. 3- the property values can be different at different location in the device. 4- the total energy content in the control volume remains unchanged. 5- the rate of energy coming into the system equals to the rate of energy exits the system.arrow_forwardWhen a hydrocarbon fuel is burned, almost all of the carbon in the fuel burns completely to form CO2 (carbon dioxide). On average, 0.59 kg of CO2 is produced for each kWh of electricity generated from a power plant that burns natural gas. A typical new household refrigerator uses about 700 kWh of electricity per year. There are 50,000 households in the city. If the area of the city is 190 miles2, and if all that CO2 accumulates in the atmosphere, calculate the increase in CO2 concentration by weight in the atmosphere knowing that each cm2 of area carries a column of air that weighs 1 kg. If 1 mature tree absorbs 48 lbm of CO2 per year, what is the ratio of trees to households this city should have to counter the effect of refrigeration?arrow_forwardThe Helmholtz free energy of a liquid column that rises, due to surface tension, inside a capillary tube (Figure 1), as a function of the height, h, is given by: F(h) =(ro)gr^2h^2/2 = -2 pi sigma r h cos teta, where r is the radius of the tube, g is the local ac- celaration of gravity, is the surface tension of the liquid, and is the contact angle of the liquid in contact with the wall of the tube. (a) From this expression, obtain the value of h as a function of the other physical parameters at equilibrium.Calculate the height that water will rise in a capil-lary of diameter 0.05mm. Assume that the contactangle between the water and the tube is zero. Thesurface tension of water at experimental conditionsisσ= 7.73×10−2N/m, and the local accelarationof gravity isg= 9.7m/s2.arrow_forward
- Saponification is a process wherein soap is added to a certain type of oil to produce grease. One such grease is said to have 75.7 % by volume oil and 24.3% by volume soap, wherein the oil and soap have densities 760 kgm/m3 and 6,250 kgm/m3, respectively. This kind of grease is sold by packs shaped like a sphere with a diameter of 20 cm for convenience. Calculate: a) the mass of oil per pack (kgm), b) the mass of soap per pack (kgm), c) the weight of each pack (kgf), d) the specific weight of grease (N/m3), e) will the grease float if it is placed in water whose density is 1000 kgm/m3? Explain.arrow_forwardWhen a hydrocarbon fuel is burned, almost all of the carbon in the fuel burns completely to form CO2 (carbon dioxide). On average, 0.59 kg of CO2 is produced for each kWh of electricity generated from a power plant that burns natural gas. A typical new household refrigerator uses about 700 kWh of electricity per year. The city's refrigerators produce. 20,650 mton CO2/year. If the area of the city is 190 miles2, and if all that CO2 accumulates in the atmosphere, calculate the increase in CO2 concentration by weight in the atmosphere knowing that each cm2 of area carries a column of air that weighs 1 kg. If 1 mature tree absorbs 48 lbm of CO2 per year, what is the ratio of trees to households this city should have to counter the effect of refrigeration?arrow_forwardA nuclear power station is situated in Coal Valley, which is a roughly rectangular valley that is 5 km long, 2 km wide, and 200 m deep. You have been asked to evaluate the effects of a worst-case scenario where the reactor housing fails, and radiation is released to the atmosphere. In your evaluation, you determine that 120 kg of Iodine-131 (a radioisotope that causes thyroid gland and liver damage) could be released into the atmosphere. Assuming the release of Iodine-131 was very rapid and all of it was uniformly distributed through the valley’s atmosphere with none escaping the valley, what would the concentration of Iodine-131 be in the valley’s air? Your answer should be expressed in units of ppm(v), and you may assume an atmospheric pressure of 1.0 atm and a temperature of 20oC. 2. Assuming the Iodine-131 concentration you calculated in part (a) is the initial concentration in the valley, you now want to determine the time it will take for the concentration to decrease to…arrow_forward
- A 5-feet diameter Chimney is designed to handle a flue gas produced in a steam power plant at a rate of 17 lb/s. The barometric pressure is 29.92-inch Hg. Outside air enters the combustion chamber at 90 F. The average temperature of the flue gas inside the Chimney is 572 F and its molecular weight is 30. Cv = 0.35, Rair = 53.342 ft-lb/lb-R. Calculate: 5. The total draft, feet of air. 6. The height of the Chimney, feetarrow_forwardHow does the average velocity of pedestrians in a city vary with the population size?arrow_forwardHeat transfer principles of convectionarrow_forward
- the title of the book: Engineering Thermofluids Thermodynamics, Fluid Mechanics, and Heat Transfer by: Mahmoud Massoudarrow_forwardDetermine the maximum temperature required for the water of 1.7 liters to prepare Arabic coffee for a family using the following data. - Heat energy of 622 kJ is supplied to the water - Specific heat capacity of water is 4.19 kJ/kg K - Density of water as 999.4 kg/m3 - Room temperature is 23 C a) Mass of water (in kg) b) Maximum temperature requiredarrow_forwardNewton's Law of Cooling - Differential Equations The rate at which a body cools is proportional to the difference in temperature between the body and its surroundings. If a body in air at 0°C will cool from 200°C to 100°C in 40 minutes, how many more minutes will it take the body to cool from 100°C to 50°C?arrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





