
Concept explainers
Draw the structure for each of the following:
- a. isobutyraldehyde
- b. diisopentyl
ketone - c. 3-methylcyclohexanone
- d. 2,4-pentanedione
- e. 4-bromo-3-heptanone
- f. 4-bromohexanal
(a)
Interpretation:
The structure of Isobutyraldehyde has to be drawn.
Concept Introduction:
For any given organic compound name, the corresponding structure can be analysed and drawn by analysing or identifying the following aspects of the name:
- Parent chain.
- Functional group.
- Position of the functional group on the parent chain.
- Alkyl substituent.
- Position of the alkyl substituent on the parent chain.
Answer to Problem 28P
The structure of Isobutyraldehyde is given below:
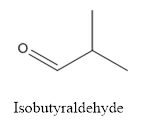
Explanation of Solution
The compound has aldehyde group that is attached to the isobutyl group.
The structure of isobutyraldehyde is as follows.

(b)
Interpretation:
The structure of diisopentyl ketone has to be drawn.
Concept Introduction:
For any given organic compound name, the corresponding structure can be analysed and drawn by analysing or identifying the following aspects of the name:
- Parent chain.
- Functional group.
- Position of the functional group on the parent chain.
- Alkyl substituent.
- Position of the alkyl substituent on the parent chain.
Answer to Problem 28P
The structure of diisopentyl ketone is given below:
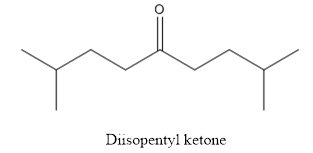
Explanation of Solution
The compound has ketone functional group. Two isopentyl groups are attached to the keto group.
The structure of diisopentyl ketone is as follows.

(c)
Interpretation:
The structure of 3-methylcyclohexanone has to be drawn.
Concept Introduction:
For any given organic compound name, the corresponding structure can be analysed and drawn by analysing or identifying the following aspects of the name:
- Parent chain.
- Functional group.
- Position of the functional group on the parent chain.
- Alkyl substituent.
- Position of the alkyl substituent on the parent chain.
Answer to Problem 28P
The structure of 3-methylcyclohexanoneis given below:
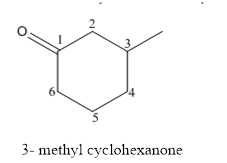
Explanation of Solution
The compound has ketone functional and group and it is cyclohexanone that has one methyl group at 3rd position of the cyclic ring.
The structure of 3-methyl cyclohexanone is as follows.

(d)
Interpretation:
The structure of 2,4-pentanedione has to be drawn.
Concept Introduction:
For any given organic compound name, the corresponding structure can be analysed and drawn by analysing or identifying the following aspects of the name:
- Parent chain.
- Functional group.
- Position of the functional group on the parent chain.
- Alkyl substituent.
- Position of the alkyl substituent on the parent chain.
Answer to Problem 28P
The structure of 2,4-pentanedione is given below:
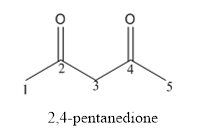
Explanation of Solution
The compound has two keto functional groups and these are attached to the pentane carbon chain.
The structure of 2,4-pentanedione is as follows:

(e)
Interpretation:
The structure of 4-bromo-3-heptanone has to be drawn.
Concept Introduction:
For any given organic compound name, the corresponding structure can be analysed and drawn by analysing or identifying the following aspects of the name:
- Parent chain.
- Functional group.
- Position of the functional group on the parent chain.
- Alkyl substituent.
- Position of the alkyl substituent on the parent chain.
Answer to Problem 28P
The structure of 4-bromo-3-heptanone is given below:
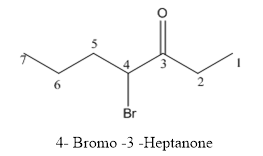
Explanation of Solution
The compound has keto functional group and it is attached to the heptane carbon chain. At the 4th position of the carbon chain has bromine attachment.

(f)
Interpretation:
The structure of 4-bromohexenal has to be drawn.
Concept Introduction:
For any given organic compound name, the corresponding structure can be analysed and drawn by analysing or identifying the following aspects of the name:
- Parent chain.
- Functional group.
- Position of the functional group on the parent chain.
- Alkyl substituent.
- Position of the alkyl substituent on the parent chain.
Answer to Problem 28P
The structure of 4-bromohexenal is given below:
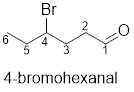
Explanation of Solution
The compound has bromine functional group at the 4th position from the aldehyde functional group on the hexane parent chain.
The structure of 4-bromohexenal is as follows:

Want to see more full solutions like this?
Chapter 12 Solutions
Essential Organic Chemistry Study Guide & Solution Manual, Books a la Carte Edition
- Figure 13.13 focuses on the use of thiol chemistry by skunks. If thiols are more volatile than alcohols and less soluble in water, why do these properties represent an advantage to skunks?arrow_forwardA stockroom assistant prepares three bottles, each containing one of the following compounds: However, the bottles are left unlabeled. Instead of discarding the samples, another assistant runs tests on the contents, using Tollens and Benedicts reagents. Match the compound to the bottle, based on these results. Tollens Reagent Benedicts Reagent Bottle A A dark precipitate forms A red-orange precipitate forms Bottle B No reaction No reaction Bottle C A dark precipitate and slight mirror form No reactionarrow_forwardDraw the organic products formed when attached allylic alcohol A is treated with following reagent. PCCarrow_forward
- The three-carbon diol used in antifreeze is It is nontoxic and is used as a moisturizing agent in foods. Oxidation of this substance within the liver produces pyruvic acid, which can be used by the body to supply energy. Give the structure of pyruvic acid.arrow_forwardReview the oxidation reactions using Cr6+reagents. Then draw the product formed when compound B is treated with following reagent. PCCarrow_forwardCyclohexane is expected to be most soluble in: HCl water None of the above pentane HIarrow_forward
- i. Why is the boiling point of the aldehyde greater than that of the alkane? ii. Why is the boiling point of alcohol the highest? iii. Explain why the solubility of aldehydes and alcohols falls as the molecules get bigger.arrow_forwardComplete each of the following reactions: a. b.arrow_forwardAnhydrous oxidations of primary alcohols can stop the oxidation at the aldehyde stage. A common reagent is Pyridinium chlorochromate (PCC). Draw the expected oxidation product of a primary alcohol with PCC.arrow_forward
- What is the difference between Tollens reagent and other oxidizing agents? What isits importance? Write the balanced equation of the sample that gave positive test forTollens test from the picture belowarrow_forward(a) Write a chemical test to distinguish between: (i) Chlorobenzene and Benzyl chloride. (ii) Chloroform and Carbon tetrachloride. (b) Why is methyl chloride hydrolysed more easily than chlorobenzene?arrow_forwardPhosgene (COCl2) was used as a poison gas in World War I. Draw the product that would be formed from the reaction of phosgene with excess of methylamine.arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning  Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





