
(a)
Interpretation:
The relationship between the given isomers needs to be established.
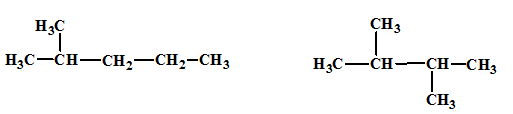
Concept Introduction:
Isomers are two or more compounds which have the same formula but different structures and properties. Constitutional isomers are structural isomers which have the same molecular formula but different connectivity of the constituent atoms.
(b)
Interpretation:
The relationship between the given isomers needs to be established.
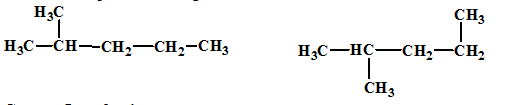
Concept Introduction:
Isomers are two or more compounds which have the same formula but different structures and properties. Constitutional isomers are structural isomers which have the same molecular formula but different connectivity of the constituent atoms.
(c)
Interpretation:
The relationship between the given isomers needs to be established.
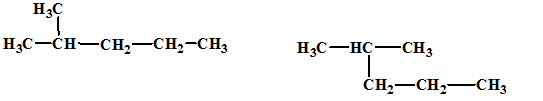
Concept Introduction:
Isomers are two or more compounds which have the same formula but different structures and properties. Constitutional isomers are structural isomers which have the same molecular formula but different connectivity of the constituent atoms.
(d)
Interpretation:
The relationship between the given isomers needs to be established.
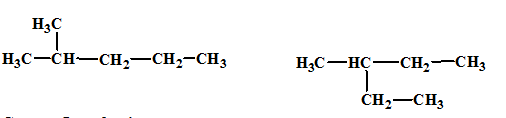
Concept Introduction:
Isomers are two or more compounds which have the same formula but different structures and properties. Constitutional isomers are structural isomers which have the same molecular formula but different connectivity of the constituent atoms.
(e)
Interpretation:
The relationship between the given isomers needs to be established.
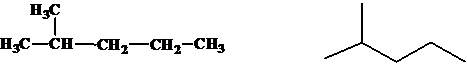
Concept Introduction:
Isomers are two or more compounds which have the same formula but different structures and properties. Constitutional isomers are structural isomers which have the same molecular formula but different connectivity of the constituent atoms.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
CONNECT IA GENERAL ORGANIC&BIO CHEMISTRY
- Among the following organic compounds the only compound which is an alcohol is C2H5OH,CH3CHO,C2H6,CH3COCH3 O CH3CHO O CH6 C2H, O C2H5OH OCH3COCH3arrow_forwardDraw the structural formula for 3-chlorohexanenitrile. O O O O O CT CH 3 CH ₂ CH CH ₂ CH ₂ CN CH 3 CH ₂ CH ₂ CH CH ₂ CH ₂ CN CI CH3 CH₂ CH₂ CH2 CH CH ₂ CN CI CH3 CH2 CH₂ CH CH ₂ CN दा CH3 CH ₂ CH CH ₂ CH ₂ CH ₂ CNarrow_forwardName the following compounds. QH CH3 | CICH₂ CH CH₂CH ČCH 3 OH O CH Draw the condensed structural formula, 3-chloro-4-oxo hepaNal cis-2, 4-dimethyl cyclohexanoNearrow_forward
- Which isomers contain an isopropyl group?arrow_forwarda) Draw the structures of the following organic compounds. i) cis-pent-2-ene ii) o-difluorobenzene iii) 3-phenylhexanoic acid b) Determine the IUPAC name of the following organic compounds. H H₂C- CH- pat. CH -CH₂ CI CI CHỊU CÁCHỊ CHỊCH, -CH₂ CH₂ CH₂ H-N CH₂ CH₂ CH₂ CH₂ c) Determine and draw the structure of the organic product for each reaction below. Br A) + NH3 B) B) C=C-CH3 + HCI H Harrow_forward< Question 20 of 45 Identify the relationship between (2R,4S)-2-bromo-4-chlorohexane and (2S,4R)-4-bromo-2-chlorohexane. A) enantiomers B) diastereomers C) geometric isomers Submit D) constitutional isomersarrow_forward
- Give the IUPAC name for the organic compound shown here: CH3CHBrCH(CH3)CH2CH3arrow_forwardWrite the name of the following compounds? 1)CH3-CH2-CH2-O-CO-CH2-CH3 2) 2,2,3-trimethyl-1-heptanolarrow_forward3 Write the structural formulas of the alcohols that must be oxidized to give chemical compounds with the given semi-structural formulas. a) H3C-CH,-CH-CHOarrow_forward
- Complete the following molecular formula of Ethyl acrylate with C, CH, CH2, or CH3.arrow_forwardFive structural isomers, or constitutional isomers, have the formula C6H14C6H14. Draw the indicated isomers, grouped by number of carbon atoms in the main chain. a) Draw two isomers with 5 carbon atoms in the main chain. Be sure to include all hydrogen atoms. b) Draw two isomers with 4 carbon atoms in the main chain. Be sure to include all hydrogen atoms.arrow_forwardName the following alcohol. OH H3C CH3 CH3 CH3 ☑arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,





