
Concept explainers
(a)
Interpretation:
The number of C atoms present in the longest chain of the molecule shown below needs to be deduced.
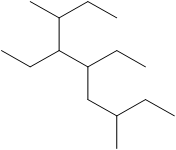
Concept Introduction:
(a)
Interpretation:
The number of C atoms present in the longest chain of the molecule shown below needs to be deduced
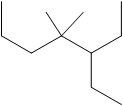
Concept Introduction:
Alkanes have a general formula, CnH2n+2where n denotes the number of C atoms. In the case of linear or branched alkanes, the parent chain is the one which has the highest number of carbon atoms. The longest continuous chain of C atoms forms the base name of the compound
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
CONNECT IA GENERAL ORGANIC&BIO CHEMISTRY
- How many electron pairs are shared when a triple bond exists between two carbon atoms? What must he the geometric arrangement around the carbon atoms in a triple bond? Draw the Lewis structure of a simple molecule that contains a triple bond.arrow_forwardAre the pairs of compounds shown below the same molecule, isomers, or different molecules?arrow_forwardExplain the different types of Hydrocarbons ?arrow_forward
- How many carbons are present in this structure? fuxarrow_forwardHow many hydrogen atoms are in 2,2-dimethylhexane?arrow_forward4: Draw out the abbreviated structural formula of the following molecules. 4-Methyl-2-hexene 2-Methyl-2-hexene 2-Hexene 5: Draw the structure of 2-Butene, labeling the n bond and all the B bonds. 6: Draw all the structures possible with the formula CSH8.arrow_forward
- How many hydrogen atoms are in an alkane moleculewith nine carbon atoms? How many are in an alkenewith nine carbon atoms and one double bond?arrow_forwardIn terms of organic chemistry how do you know if an atom has ionic bonds or covalent bonds and how do you know if it has just one of these or if it has both? Could you provide some examples of what an ionic bond looks like, covalent bond looks like, and what an atom would look like with both of these?arrow_forwardWhat are the key differences in the way that alkanes, alkenes, and alkynes are named?<arrow_forward
- How many chiral carbons does this molecule contain?arrow_forwardDraw the expanded structural formula for 1,3-dichlorocyclopentane.An expanded structural formula shows all the atoms of the molecule and all the bonds between the atoms in the molecule.arrow_forwardAlkenes are unsaturated hydrocarbons because they have fewer thanthe maximum number of hydrogen atoms per carbon. Define this ?arrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHERChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHERChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





