
Concept explainers
(a)
Interpretation:
The electron dot formula and structural formula for
Concept introduction:
The electron dot formula shows the valence electrons which form the bond between the atoms in a molecule. The electron pairs that are shared by the atoms are known as bonding electrons. The other electrons that are present in order to complete the octet are known as non-bonding electrons. The electron dot formula is also known as the Lewis structure.
Answer to Problem 35E
The electron dot formula for
![]()
The structural formula for
![]()
Explanation of Solution
The total number of valence electron in
![]()
Figure 1
Out of five electron pairs, three electron pairs are involved in bonding. Therefore, there is two lone pair. The structural formula for
![]()
Figure 2
The electron dot formula for
The structural formula for
(b)
Interpretation:
The electron dot formula and structural formula for
Concept introduction:
The electron dot formula shows the valence electrons which form the bond between the atoms in a molecule. The electron pairs that are shared by the atoms are known as bonding electrons. The other electrons that are present in order to complete the octet are known as non-bonding electrons. The electron dot formula is also known as the Lewis structure.
Answer to Problem 35E
The electron dot formula for
![]()
The structural formula for
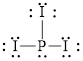
Explanation of Solution
The total number of valence electron in
![]()
Figure 3
Out of thirteen electron pairs, only three electron pair is involved in bonding. Therefore, there is ten lone pair. The structural formula for

Figure 4
The electron dot formula for
The structural formula for
(c)
Interpretation:
The electron dot formula and structural formula for
Concept introduction:
The electron dot formula shows the valence electrons which form the bond between the atoms in a molecule. The electron pairs that are shared by the atoms are known as bonding electrons. The other electrons that are present in order to complete the octet are known as non-bonding electrons. The electron dot formula is also known as the Lewis structure.
Answer to Problem 35E
The electron dot formula for
![]()
The structural formula for
![]()
Explanation of Solution
The total number of valence electron in
![]()
Figure 5
Out of nine electron pairs, four electron pair is involved in bonding. Therefore, there is five lone pair. The structural formula for
![]()
Figure 6
The electron dot formula for
The structural formula for
(d)
Interpretation:
The electron dot formula and structural formula for
Concept introduction:
The electron dot formula shows the valence electrons which form the bond between the atoms in a molecule. The electron pairs that are shared by the atoms are known as bonding electrons. The other electrons that are present in order to complete the octet are known as non-bonding electrons. The electron dot formula is also known as the Lewis structure.
Answer to Problem 35E
The electron dot formula for
![]()
The structural formula for
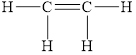
Explanation of Solution
The total number of valence electron in
![]()
Figure 7
All electron pair is involved in bonding. Therefore, there is no lone pair. The structural formula for

Figure 8
The electron dot formula for
The structural formula for
Want to see more full solutions like this?
Chapter 12 Solutions
Modified Mastering Chemistry with Pearson eText -- Standalone Access Card -- for Introductory Chemistry: Concepts and Critical Thinking (8th Edition)
- Which atoms can bond to sulfur so as to produce a positive partial charge on the sulfur atom?arrow_forward3-25 Why are carbon and silicon reluctant to form ionic bonds?arrow_forwardin the molecule PCI3 given the phosphorus has the electronegativity of 2.1 and the chlorine has an electronegativity of three point no we can conclude that this molecule isarrow_forward
- structural formula for the nitrite ion, NO2–arrow_forwardAmmonium nitrate is an inexpensive source of nitrogen content used to enrichfertilizers. Although when handled improperly, it could be dangerous andcatastrophic. What is the chemical formula for ammonium nitrate?arrow_forwardWhat are the chemical structure of the following?arrow_forward
- Write the structural formulas of 4 compounds with the formula C3H6Oarrow_forwardGive a cation isoelectronic with fluoride ion______________________ 3.Arrange in increasingsize: sodiumion, nitride ion, magnesium ion, oxide ion, neon. (explain your rationale)______<______<______<_____<_____ Arrange in decreasingsize: sulfide ion, argon, potassium ion, calcium ion, chloride ion. (explain your rationale)______>______>______>_____>___arrow_forwardWrite the chemical formula of the following. A. Calcium chloride B. Calcium chloratearrow_forward
- Give the electron dot diagram for N, O, S, H2O, and methane.arrow_forwardThe empirical formula of a covalent compound is CH2. Which of the following is not a possible chemical formula for this compound?arrow_forwardIn many ways, arsenate (AsO4 3−) is very similar to phosphate (PO4 3-), yet it does not substitute for phosphate in biomolecules. After reviewing the essential atomic characteristics of the element arsenic, explain this phenomenon.arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax

