
ORGANIC CHEMISTRY (LL) W/WILEYPLUS NEXT
12th Edition
ISBN: 9781119664635
Author: Solomons
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12, Problem 37P
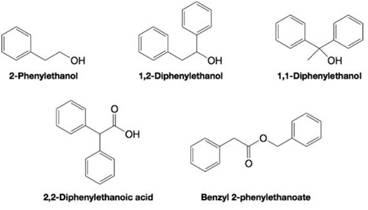
Explain how
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
How could 1H NMR spectroscopy be used to distinguish among isomers A, B, and C?
Identify the compound with molecular formula C8H10O that gives the IR and 1H NMR spectra shown here.
identify the compound with molecular formula C2H6O that gives this 1H NMR spectrum.
Chapter 12 Solutions
ORGANIC CHEMISTRY (LL) W/WILEYPLUS NEXT
Ch. 12 - Prob. 1PPCh. 12 - Prob. 2PPCh. 12 - Prob. 3PPCh. 12 - PRACTICE PROBLEM 12.4 Predict the products of the...Ch. 12 - Prob. 5PPCh. 12 - Prob. 6PPCh. 12 - Practice Problem 12.7
Provide retrosynthetic...Ch. 12 - Prob. 8PPCh. 12 - What products would you expect from the reaction...Ch. 12 - What products would you expect from the reaction...
Ch. 12 - What product (or products) would be formed from...Ch. 12 - Prob. 12PCh. 12 - 12.13 Write reaction conditions and the product...Ch. 12 - Prob. 14PCh. 12 - Predict the organic product from each of the...Ch. 12 - Predict the organic product from each of the...Ch. 12 - Predict the organic product from each of the...Ch. 12 - Predict the major organic product from each of the...Ch. 12 - Synthesize each of the following compounds from...Ch. 12 - Prob. 20PCh. 12 - 21. Write a mechanism for the following reaction....Ch. 12 - Prob. 22PCh. 12 - 23. What organic products A-H would you expect...Ch. 12 - Prob. 24PCh. 12 - Show how 1-pentanol could be transformed into each...Ch. 12 - Provide the reagents needed to accomplish...Ch. 12 - Prob. 27PCh. 12 - For each of the following alcohols, write a...Ch. 12 - Prob. 29PCh. 12 - Prob. 30PCh. 12 - Prob. 31PCh. 12 - Prob. 32PCh. 12 - Predict the major organic product from each of the...Ch. 12 - 34. Synthesize the following compound using...Ch. 12 - Prob. 35PCh. 12 - Prob. 36PCh. 12 - 37. Explain how and IR spectroscopy could be used...Ch. 12 - 38. An unknown X shows a broad absorption band in...Ch. 12 - Prob. 39PCh. 12 - The problem below is directed toward devising a...
Additional Science Textbook Solutions
Find more solutions based on key concepts
What dipeptides would be formed by heating a mixture of valine and N-protected leucine?
Organic Chemistry (8th Edition)
10.71 Identify each of the following as an acid or a base: (10.1)
H2SO4
RbOH
Ca(OH)2
HI
...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Q1. What is the empirical formula of a compound with the molecular formula
Chemistry: A Molecular Approach
For Practice 1.1
Is each change physical or chemical? Which kind of property (chemical or physical) is demonst...
Principles of Chemistry: A Molecular Approach (3rd Edition)
Name each of the following:
Organic Chemistry
1.6 Read the labels on products used to wash your dishes. What are the names of some chemicals contained in tho...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Indicate two basic differences that exist between the spectra of 1H y 13C in NMR.arrow_forward5. Determine the structure of a compound with a molecular formula of C4H8O2 with the 13C and 'H NMR spectra below. No peaks exchange with D20 on the 'H NMR spectrum. ada I=2 |=3 1=3 1.2 4.0 4.0 3.0 2.0 1.0arrow_forwardDetermine the molecular structure of the compound in the IR spectrum data, 1H NMR ; 13C NMR and MS, explain it for each picture.arrow_forward
- These are the 1H RMN and IR spectrums of an A compound with formula C10H13NO2. When A reacts with NaOH in water and heat, B compound is formed, with formula C10H11NO. What are the structures of A and B?arrow_forwardMass spectra of butylcyclopentane and tert-butylcyclopentane were acquired. Spectrum A exhibited significant mass peaks at m/z values of 126, 97, 83, 69, 55, and 41. Spectrum B exhibited significant peaks at m/z values of 111, 69,57, and 41. Match each spectrum with its compound.arrow_forwardReduction of cyclohex-2-enone can yield cyclohexanone, cyclohex-2enol, or cyclohexanol, depending on the reagent and reaction conditions. How could you use IR spectroscopy to distinguish the three possible products?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY