
Interpretation:
For each transformation, the reducing agent used, out of
Concept introduction:
Lithium aluminium hydride and sodium borohydride are reducing agents.
Answer to Problem 3PP
Solution:
(a)
(b)
(c)
(d)
(e)
(a)
Explanation of Solution
The given transformation is as follows:
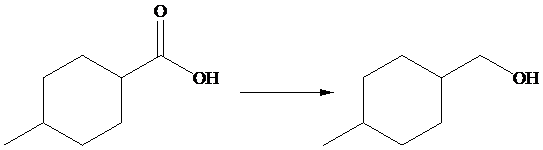
In the above transformation, carboxylic acid is converted into alcohol. This transformation is done by using
Hence,
(b)
The given transformation is as follows;
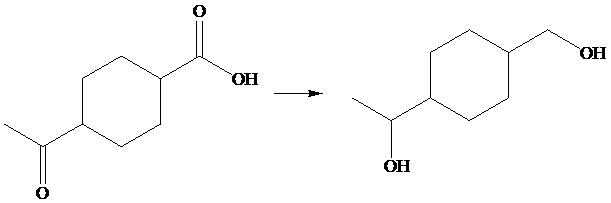
In the above transformation, carboxylic acid and ketone are converted into their corresponding alcohols. This transformation is done by using
Hence,
(c)
The given transformation is as follows:
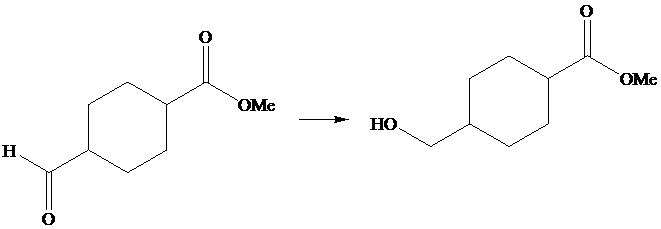
In the above transformation, aldehyde is converted into its corresponding alcohol, but there is no effect on the ester group. This transformation is done by using
Hence,
(d)
The given transformation is as follows:
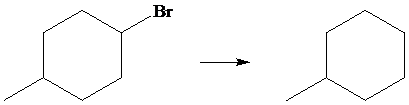
In the above transformation, the bromine atom is replaced by the hydrogen atom. This transformation is done by using
Hence,
(e)
The given transformation is as follows:
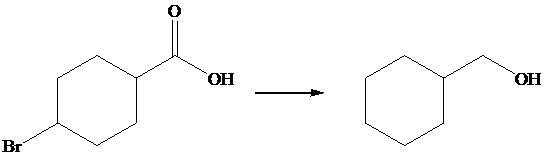
In the above transformation, the bromine atom is replaced by the hydrogen atom, and carboxylic acid is converted into its corresponding alcohol. This transformation is done by using
Hence,
Want to see more full solutions like this?
Chapter 12 Solutions
Organic Chemistry (Looseleaf) - With Access
- What are the reagents need for the structure in order to accomplish the following transformations. Explain brieflyarrow_forward#12. Outline a synthesis of the following molecules starting with ethyne and CH2I2 as the only carbon sources and any needed inorganic reagents. Obviously more than one ethyne molecule is needed for each of the syntheses.arrow_forwardSuggest and explain two synthetic methods for the conversion of compound 6 to compound 4. Include side reactionsarrow_forward
- A student was given from the list of the compounds below A, B and D blindly and asked to identify them all. He treated each of them with Brady's reagent (2,4-ditrophenylhydrazine) and isolated a bright yellow compound for one of them, but the other two gave false negatives. The student reasoned that the false negatives may be due to sterics and, on further thinking, it dawned on him that he might be able to rule out one of the false negatives with the haloform test. What compound did he find compatible with the haloform test? That compound did indeed give a false negative in the Brady test. Which of the other two was positive in the Brady test? A = haloform B = Brady A = haloform D = Brady B = haloform A = Brady B = haloform D = Brady D = haloform A = Brady D = haloform B = Bradyarrow_forwardGive the reagents and conditions necessary for the following conversion. A to B, B to D, B to C, B to E, E to F, E to G, G to H, H to I. Hence deduce the name and structural formula of the compounds C and I. Compare the procedure for converting F and E to G.arrow_forwardThis is a transesterification reaction. It mechanistically is the same as a Fischer Esterification. Write a mechanism for the reaction. Include all lone pairs and formal charges present throughout the mechanism.arrow_forward
- Show the electron-flow mechanism of the synthetic schemes (R-Salsolinol) of the picture below. This involves predicting major and by-products using electronic and structural effects. The arrow push mechanism must be shown.arrow_forwardThe following equation shows the bromination of compound 1. Propose a structure for product D and a curved arrow mechanism that accounts for its formation featuring the initiation, propagation and termination steps. can you also discuss the stereochemical outcome.arrow_forwardPlease show the electron-flow mechanism of the general synthesis of Benzyl Chloride from Phenol. This involves predicting major and by-products using electronic and structural effects. The arrow push mechanism must be shown.arrow_forward
- Conversion of an alkene to a halohydrin and internal displacement of a halide ion by an alkoxide ion are both stereoselective. Use this information to demonstrate that the configuration of the alkene is preserved in the epoxide. As an illustration, show that reaction of cis-2-butene by this two-step sequence gives cis-2,3- dimethyloxirane (cis-2-butene oxide).arrow_forwardCompound X gives good recrystalisation results using the following three solvents ethanol,methanol,and dichloromethane .Which solvent will you select as the best solvent out of those three and why?arrow_forwardwe know that ethers, such as diethyl ether and tetrahydrofuran, are quite resistant to the action of dilute acids and require hot concentrated HI or HBr for cleavage. However, acetals in which two ether groups are linked to the same carbon undergo hydrolysis readily, even in dilute aqueous acid. How do you account for this marked difference in chemical reactivity toward dilute aqueous acid between ethers and acetals?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





