
Concept explainers
Interpretation:
Boiling point of water and ethyl alcohol must be found at 1 atm from the given figure.
Also, the boiling point of water must be estimated at 0.8 atm pressure.
Concept introduction:
Boiling point of a liquid is the temperature at which vapor pressure of the liquid is equal to the atmospheric pressure.
Answer to Problem 90A
Boiling point of water at 1 atm is 100°C.
Boiling point of ethyl alcohol at 1 atm pressure is 78°C.
Boiling point of water at 0.8 atm is approximately 96°C.
Explanation of Solution
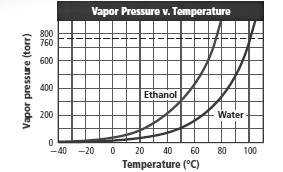
To get the boiling point the corresponding temperature must be found from the graph at which the vapor pressure is equal to the atmospheric pressure (760 torr).
The graph shows that vapor pressure reaches to 760 torr for water and ethyl alcohol at 100°C and 78°C respectively.
So boiling point of water and ethyl alcohol are 100°C and 78°C respectively.
Temperature corresponding to 0.8 atm (608 torr) is 96°C. Thus estimated boiling point of water at 0.8 atm is 96°C.
Boiling point of water and ethyl alcohol are 100°C and 78C respectively.
Estimated boiling point of water at 0.8 atm is 96°C.
Chapter 12 Solutions
Glencoe Chemistry: Matter and Change, Student Edition
Additional Science Textbook Solutions
General Chemistry: Principles and Modern Applications (11th Edition)
Inorganic Chemistry
Introductory Chemistry (5th Edition) (Standalone Book)
General, Organic, and Biological Chemistry (3rd Edition)
Chemistry: The Central Science (13th Edition)
Chemistry: Structure and Properties
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





