
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
5th Edition
ISBN: 9780321967466
Author: Karen C. Timberlake
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12.2, Problem 12.9QAP
Give the IUPAC name for each of the following
a. 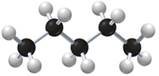
b.
c. 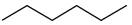
d. 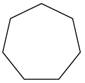
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
a volume of 448 ml of an alkane was completely burned. The combustion products were passed through an aqueous solution of calcium hydroxide, from which 8 grams of precipitate were formed. Find:
a) the molecular formula of the alkane
b) formulas of isomers of alkane structure
Using the general molecular formula for alkanes: (a) Predict the molecular formula of the C28 straight-chain alkane
Explain the role of London dispersion forces on the melting and boiling points of alkanes.
Chapter 12 Solutions
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
Ch. 12.1 - Identify each of the following as a formula of an...Ch. 12.1 - Prob. 12.2QAPCh. 12.1 - Prob. 12.3QAPCh. 12.1 - Prob. 12.4QAPCh. 12.1 - Prob. 12.5QAPCh. 12.1 - Prob. 12.6QAPCh. 12.1 - Prob. 12.7QAPCh. 12.1 - Prob. 12.8QAPCh. 12.2 - Give the IUPAC name for each of the following...Ch. 12.2 - Prob. 12.10QAP
Ch. 12.2 - Prob. 12.11QAPCh. 12.2 - Prob. 12.12QAPCh. 12.3 - Prob. 12.13QAPCh. 12.3 - Prob. 12.14QAPCh. 12.3 - Give the IUPAC name for each of the following:Ch. 12.3 - Give the IUPAC name for each of the following:Ch. 12.3 - Draw the condensed structural formula for each of...Ch. 12.3 - Draw the condensed structural formula for each of...Ch. 12.3 - Draw the line-angle structural formula for each of...Ch. 12.3 - Draw the line-angle structural formula for each of...Ch. 12.4 - Heptane, used as a solvent for rubber cement, has...Ch. 12.4 - Nonane has a density of 0.79 g/mL and boils at 151...Ch. 12.4 - Prob. 12.23QAPCh. 12.4 - Prob. 12.24QAPCh. 12.4 - Prob. 12.25QAPCh. 12.4 - Prob. 12.26QAPCh. 12.5 - Prob. 12.27QAPCh. 12.5 - Prob. 12.28QAPCh. 12.5 - Prob. 12.29QAPCh. 12.5 - Prob. 12.30QAPCh. 12.5 - Prob. 12.31QAPCh. 12.5 - Prob. 12.32QAPCh. 12.6 - 12.33 Give the IUPAC name for each of the...Ch. 12.6 - Prob. 12.34QAPCh. 12.6 - Prob. 12.35QAPCh. 12.6 - Prob. 12.36QAPCh. 12.6 - Prob. 12.37QAPCh. 12.6 - Prob. 12.38QAPCh. 12.7 - Prob. 12.39QAPCh. 12.7 - Prob. 12.40QAPCh. 12.7 - Prob. 12.41QAPCh. 12.7 - Prob. 12.42QAPCh. 12.7 - Prob. 12.43QAPCh. 12.7 - Prob. 12.44QAPCh. 12.7 - Prob. 12.45QAPCh. 12.7 - Prob. 12.46QAPCh. 12.8 - Prob. 12.47QAPCh. 12.8 - Give the IUPAC name and any common name for each...Ch. 12.8 - Prob. 12.49QAPCh. 12.8 - Prob. 12.50QAPCh. 12.8 - Prob. 12.51QAPCh. 12.8 - Prob. 12.52QAPCh. 12.8 - Prob. 12.53QAPCh. 12.8 - Prob. 12.54QAPCh. 12 - Prob. 12.55UTCCh. 12 - Prob. 12.56UTCCh. 12 - Prob. 12.57UTCCh. 12 - Prob. 12.58UTCCh. 12 - Prob. 12.59UTCCh. 12 - Prob. 12.60UTCCh. 12 - Prob. 12.61UTCCh. 12 - Prob. 12.62UTCCh. 12 - Give the number of carbon atoms and the types of...Ch. 12 - Prob. 12.64UTCCh. 12 - Prob. 12.65AQAPCh. 12 - Give the IUPAC name for each of the following:...Ch. 12 - Prob. 12.67AQAPCh. 12 - Prob. 12.68AQAPCh. 12 - Prob. 12.69AQAPCh. 12 - 12.70 Give the IUPAC name for each of the...Ch. 12 - Prob. 12.71AQAPCh. 12 - Prob. 12.72AQAPCh. 12 - Prob. 12.73AQAPCh. 12 - Prob. 12.74AQAPCh. 12 - Prob. 12.75AQAPCh. 12 - Prob. 12.76AQAPCh. 12 - Prob. 12.77AQAPCh. 12 - Prob. 12.78AQAPCh. 12 - Prob. 12.79AQAPCh. 12 - Prob. 12.80AQAPCh. 12 - Prob. 12.81AQAPCh. 12 - Prob. 12.82AQAPCh. 12 - Prob. 12.83AQAPCh. 12 - Prob. 12.84AQAPCh. 12 - Prob. 12.85AQAPCh. 12 - Prob. 12.86AQAPCh. 12 - Prob. 12.87AQAPCh. 12 - Prob. 12.88AQAPCh. 12 - Prob. 12.89CQCh. 12 - Prob. 12.90CQCh. 12 - Prob. 12.91CQCh. 12 - Prob. 12.92CQCh. 12 - Prob. 12.93CQCh. 12 - Prob. 12.94CQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Use the generic formula for alkanes (CnH2n+2) to derive molecular and condensed structural formulas for: a. Propane, 3 carbon atoms b. Octane, 8 carbon atoms c. Butane, 4 carbon atomsarrow_forwardWhen the molecular formulas for cyclic and noncyclic alkanes with the same number of carbon atoms are compared, it is always found that the cycloalkane has a) two more hydrogen atoms. b) the same number of hydrogen atoms. c) two less hydrogen atoms. d) four less hydrogen atoms.arrow_forwardUsing the general molecular formula for alkanes: Predict the molecular formula of the alkanes containing 44 carbon atoms with extensivebranchingarrow_forward
- Like alkanes, cycloalkanes are soluble in each other True or falsearrow_forward259.091 g of an unknown alkane is burned in excess oxygen. The combustion process produced 800 g of carbon dioxide. Experiment shows that for each mole of the unknown alkane us burned, 8 moles of carbon dioxide is produced. Determine the name of the alkane.arrow_forwardCompare the reactivity of alkanes and Alkenes.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning



Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License