
ORGANIC CHEMISTRY (LOOSELEAF)
6th Edition
ISBN: 9781260475630
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12.3, Problem 2P
What
a.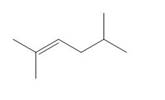 b.
b. 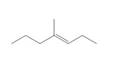 c.
c. 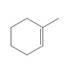
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw the structure of an alkane that:
a. Contains only 1° and 4° carbons. c. Contains only 1° and 2° hydrogens.
b. Contains only 2° carbons. d. Contains only 1° and 3° hydrogens.
c. CH2-CH2-CH2-CH-CH3
b. C
CH3
CH3
d. CH3-CH2-CH-CH,-CH--CH3
CH3
111s
C.
CH3
12.36 Give the IUPAC name for each of the follov ing
d.
alkanes.
a. CH3-CH,--CH-CH2-CH,-CH3
ČH3
12.39
b. CH3--CH2-CH2-CH-CH3
ČH3
CH
c. CH-CH2-CH-CH3
ČH3
CH3
d. CH3-CH-CH2-CH-CH3
CH3
CH3
14.4
12.37 Give the IUPAC name for each of the followi ng
alkanes.
a. CH3-CH-CH2-CH-CH-CH3
CH3
CH3 CH3
CH3
b. CH3--C-CH,-CH-CH3
CH3
CH3
12
CH3
c. CH3-CH2-C-CH2-CH3
CH2
CH3
CH3
d. CH3-CH2-C-CH2-CH2
CH3
CH2
1.
CH3
12.38 Give the IUPAC name for each of the follo ng
alkanes.
a. CH3-CH2-CH-CH-CH-CH-C
CH3 CH2 CH3
CH3
Question 1: What alkane is formed when each alkene is treated with H2 and a Pd
catalyst?
a.
CH3
C=C
CH₂CH(CH3)2
CH3 H
b.
C.
Chapter 12 Solutions
ORGANIC CHEMISTRY (LOOSELEAF)
Ch. 12.1 - Prob. 1PCh. 12.3 - Problem 12.2 What alkane is formed when each...Ch. 12.3 - Prob. 3PCh. 12.3 - Prob. 4PCh. 12.3 - Prob. 5PCh. 12.3 - Prob. 6PCh. 12.3 - Compound Molecular formula before...Ch. 12.4 - Problem 12.8 Draw the products formed when...Ch. 12.5 - Prob. 9PCh. 12.5 - Prob. 10P
Ch. 12.5 - Problem 12.11 (a) Draw the structure of a compound...Ch. 12.5 - Prob. 12PCh. 12.5 - Prob. 13PCh. 12.6 - Problem 12.14 Draw the products of each...Ch. 12.8 - Prob. 15PCh. 12.8 - Problem 12.16 Draw all stereoisomers formed when...Ch. 12.9 - Prob. 17PCh. 12.9 - Problem 12.18 Draw the products formed when both...Ch. 12.10 - Problem 12.19 Draw the products formed when each...Ch. 12.10 - Prob. 20PCh. 12.10 - Prob. 21PCh. 12.11 - Problem 12.22 Draw the products formed when each...Ch. 12.11 - Prob. 23PCh. 12.12 - Problem 12.24 Draw the organic products in each of...Ch. 12.13 - Prob. 25PCh. 12 - 12.29 Draw the products formed when A is treated...Ch. 12 - Prob. 30PCh. 12 - Prob. 31PCh. 12 - Prob. 32PCh. 12 - Prob. 33PCh. 12 - Draw the organic products formed when cyclopentene...Ch. 12 - Draw the organic products formed when allylic...Ch. 12 - Prob. 39PCh. 12 - Prob. 40PCh. 12 - Prob. 41PCh. 12 - What alkene is needed to synthesize each 1,2-diol...Ch. 12 - 12.48 Draw the products formed in each oxidative...Ch. 12 - What alkene or alkyne yields each set of products...Ch. 12 - Prob. 47PCh. 12 - Prob. 48PCh. 12 - Prob. 49PCh. 12 - Prob. 50PCh. 12 - 12.57 Draw the product of each asymmetric...Ch. 12 - 12.60 Identify A in the following reaction...Ch. 12 - Prob. 58PCh. 12 - 12.62 It is sometimes necessary to isomerize a cis...Ch. 12 - 12.63 Devise a synthesis of each compound from...Ch. 12 - Prob. 61P
Additional Science Textbook Solutions
Find more solutions based on key concepts
Describe the orbitals used in bonding and the bond angles in the following compounds: a. CH3O b. CO2 c. H2CO d....
Organic Chemistry (8th Edition)
Which of the following solutions has the higher molarity? 10 ppm KI in water or 10,000 ppb KBr in water 0.25 ma...
CHEMISTRY-TEXT
Practice Problem ATTEMPT
Write the rate expressions for each of the following reactions:
(a)
(b)
(c)
Chemistry
Give the IUPAC name for each compound.
Organic Chemistry
The active ingredient in Tylenol and a host of other over-the-counter pain relievers is acetaminophen (C8H9NO2)...
Chemistry: Atoms First
Characterize each of the following structures as aromatic, nonaromatic, or antiaromatic:
Answer: _____
Organic Chemistry As a Second Language: Second Semester Topics
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Glycerol contains: a. oxygens which are each bonded to two alkyl groups b. oxygens single-bonded to primary and secondary carbons c. Oxygens double-bonded to carbon, with alkyls on both sides d. Oxygens double-bonded to carbon, with alkyls on one side only e. Oxygens double-bonded to carbon, with an alkyl on one side and an --OH on the other sidearrow_forwardGive the IUPAC name for each. a. I have 5-bromo-3methylheptanearrow_forwardGive an IUPAC name for the following as hydrocarbons with the benzene ring as a substituent: a. b.arrow_forward
- Draw the structure of an alkane that: c. contains only 1° and 2° hydrogens d. contains only 1° and 3° hydrogens a. contains only 1° and 4° carbons b. contains only 2° carbonsarrow_forwardDraw a complete structure for each alkene or alkyne. a. CH2 CHCH2CH3 b. CH3C CCH2CH3arrow_forwardDraw the products of combustion of each alkane. a. CH;CH,CH,CH2CH(CH3)2 b.arrow_forward
- Consider the structure of cyclohexene, if it undergoes epoxidation followed by exposure to water, which of the following final product is formed? a. Cyclohexan-1,2,-diol b. Cyclohexane c. Cyclohexanone d. Hexan-1,-6-dioic acidarrow_forward1. Which functional groups are present in the following molecule? HO N- Alkene, ketone, amine, alcohol, ester Alkene, ketone, alcohol, ether Alkene, amine, phenol, ether Ether, phenol, alkene, amide а. b. с. d. Which functional groups are present in the following molecule (aside from cycloalkane skeleton)? 2. он Alkene, ester, tertiary alcohol. Alkene, ether, secondary alcohol. Alkene, ketone, secondary alcohol. Alkyne, ketone, secondary alcohol. а. b. с. d.arrow_forwardWhich of the following is not a physical property of alcohols or phenols? A. The solubilities of primary alcohols in water decrease with increasing molecular weight. B. Due to hydrogen bonding, boiling points of alcohols is much higher than those of corresponding alkanes. C. The hydroxyl group of an alcohol is nonpolar. D. Phenols are generally only slightly soluble in water.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License