
Interpretation:
The condensed formulas for two
Concept introduction:
Isomer: A molecule having same molecular formula with different chemical formula is called isomer.
Condensed structure:
Condensed structure is the line diagram, the carbon-carbon bonds are represented with lines, and hydrogens are absent entirely.
Carboxylic acid:
A carbon atom is double-bonded to an oxygen atom
If the carbonyl carbon is attached with hydroxyl group is called as carboxylic acid.
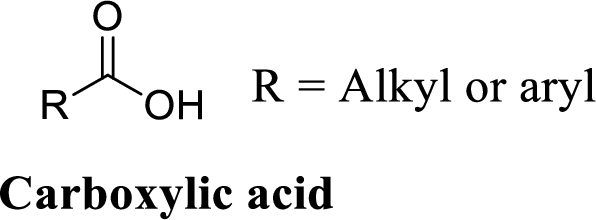
Trending nowThis is a popular solution!

Chapter 1 Solutions
OWL V2 with MindTap Reader and Student Solutions Manual eBook for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
- There are two different carboxylic acids with the formula C4H8O2. Draw and name them.arrow_forwardDraw the condensed structural formula for a carboxylic acid that has the formula C6H12O2, with no substituents.arrow_forwardWrite structural formulas for three ketones that have the formula C5H10O.arrow_forward
- Draw a condensed structural formula for the single carboxylic acid with molecular formula C3H6O2.arrow_forwardname and draw the structural formulas for the 4 carboxylic acids with the molecular formula C5H10O2arrow_forwardThere are two different butanoic acids with the formula C5H10O2. Draw and name them.arrow_forward
- Draw the expanded structural formula for a carboxylic acid that has the formula C5H10O2C5H10O2, with two methyl substituents.arrow_forwardDraw the structural formulas for the six ketones with the molecular formula C6H12O.arrow_forwardDraw Lewis structure for an isomer of C3H6O2 that is a carboxylic acid, and draw another Lewis structure for an isomer of C3H6O2 that is an ester.arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning



