
Concept explainers
Interpretation: The graph of carbon-to-hydrogen in carboxylic acid for the increasing number of carbon is to be plotted and compared with that of
Concept introduction: The general formula for the carboxylic acid is
The general formula for the alkene is
The general formula for the alkane is
To determine: The plot of carbon-to hydrogen ratio and its comparison with that of alkane and alkene.
Answer to Problem 13.104QP
The plot of carbon to hydrogen ratio in carboxylic acid correlates better with the plot of carbon to hydrogen in alkenes.
Explanation of Solution
The molecular formula of carboxylic acid containing one to six carbons, the number of carbon and hydrogen atoms and its ratio is,
Table 1
From the above data it is concluded that the general formula for the carboxylic acid is
The plot of carbon-to-hydrogen ratio in carboxylic acid as a function of number of carbon atoms is,
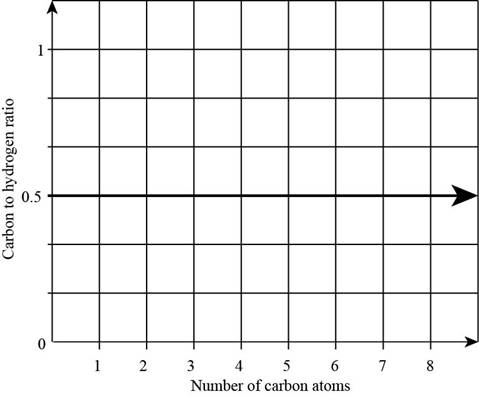
Figure 1
The general formula for the alkene is
The molecular formula of alkene containing one to six carbons, the number of carbon and hydrogen atoms and its ratio is,
Table 2
From the above data it is concluded that the carbon to hydrogen ratio in alkene is
The general formula for the alkane is
The molecular formula of alkane containing one to six carbons, the number of carbon and hydrogen atoms and its ratio is,
Table 3
From the above data it is concluded that the carbon to hydrogen ratio in alkane changes as the numbers of carbon and hydrogen atom increases.
Therefore, the plot of carbon to hydrogen ratio in carboxylic acid correlates better with the plot of carbon to hydrogen in alkenes.
The plot of carbon to hydrogen ratio in carboxylic acid correlates better with the plot of carbon to hydrogen in alkenes.
Want to see more full solutions like this?
Chapter 13 Solutions
Chemistry
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





