
Concept explainers
What molecular ion is expected for each compound?
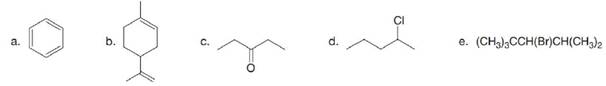
(a)
Interpretation: The expected molecular ion for the given compound is to be determined.
Concept introduction: Molecular mass is the sum of the atomic weights of each constituent element multiplied by the number of atoms of that element. The molecular weight of compound is equal to the mass of the molecular ion.
Answer to Problem 13.24P
The structure of the given compound is,
The expected molecular ion peak of
Explanation of Solution
The molecular formula of given compound is
The
The molecular weight of the compound is equal to the mass of the molecular ion.
Hence, the expected molecular ion peak of
The expected molecular ion peak of
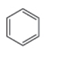
(b)
Interpretation: The expected molecular ion for the given compound is to be determined.
Concept introduction: Molecular mass is the sum of the atomic weights of each constituent element multiplied by the number of atoms of that element. The molecular weight of compound is equal to the mass of the molecular ion.
Answer to Problem 13.24P
The structure of the given compound is,
The expected molecular ion peak of
Explanation of Solution
The molecular formula of given compound is
The
The molecular weight of the compound is equal to the mass of the molecular ion.
Hence, the expected molecular ion peak of
The expected molecular ion peak of
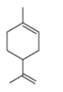
(c)
Interpretation: The expected molecular ion for the given compound is to be determined.
Concept introduction: Molecular mass is the sum of the atomic weights of each constituent element multiplied by the number of atoms of that element. The molecular weight of compound is equal to the mass of the molecular ion.
Answer to Problem 13.24P
The structure of the given compound is,
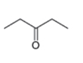
The expected molecular ion peak of
Explanation of Solution
The molecular formula of given compound is
The
The molecular weight of the compound is equal to the mass of the molecular ion.
The expected molecular ion peak of
The expected molecular ion peak of
(d)
Interpretation: The expected molecular ion for the given compound is to be determined.
Concept introduction: Molecular mass is the sum of the atomic weights of each constituent element multiplied by the number of atoms of that element. The molecular weight of compound is equal to the mass of the molecular ion.
Answer to Problem 13.24P
The structure of the given compound is,
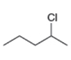
The larger molecular ion
Explanation of Solution
The molecular formula of given compound is
The
The
The molecular weight of the compound is equal to the mass of the molecular ion.
Hence, the larger molecular ion
The larger molecular ion
(e)
Interpretation: The expected molecular ion for the given compound is to be determined.
Concept introduction: Molecular mass is the sum of the atomic weights of each constituent element multiplied by the number of atoms of that element. The molecular weight of compound is equal to the mass of the molecular ion.
Answer to Problem 13.24P
The structure of the given compound is,
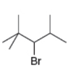
The larger molecular ion
Explanation of Solution
The molecular formula of given compound is
The
The
The molecular weight of the compound is equal to the mass of the molecular ion.
Hence, the larger molecular ion
The larger molecular ion
Want to see more full solutions like this?
Chapter 13 Solutions
ORGANIC CHEMISTRY-STUDY GDE...-W/ACCESS
- The so-called nitrogen rule states that if a compound has an odd number of nitrogen atoms, the value of m/z for its molecular ion will be an odd number. Why?arrow_forwardWhich elements on the periodic table (other than H) are likely to form a+1 cation?arrow_forwardThe chemical structure of dimethyl ether ((CH3)2O) is shown below. Highlight each atom that is in a methyl grarrow_forward
- Chemistry Name 3 major peaks and their bond types. What compounds do you think are present? What does this IR indicate about the purity of the product?arrow_forwardWhich of the molecules gives rise to a molecular ion with an odd value of m/z? A. Br2C8H14 B. NC8H13 C. Cl2C6H12 D. N2C7H10arrow_forwardExplain why CH3Cl has a greater dipole moment than CH3F even though F is more electronegative than Cl.arrow_forward
- For compound below, identify any polar covalent bonds, and indicate the direction of the dipole moment using the symbols δ+ and δ-:HCl H------Cl Indicate the dipole moment by typing the red atom number(s) as a comma separated list in numerical order (e.g. 1,2,3). Write "none" if there are no atoms with the corresponding symbol.arrow_forwardDescribe the type of bond represented by the dash for each of the following compounds using the definitions in Q1. Consider only the atoms to which the dash is directly connected. H–H H–CH3 H–NH2 Na–ONa Na–Br K–OH H–OH H–Br F–CH3 Q4 Draw the electron-dot formulas for the following compounds or ions and calculate the formal charges for every atom. HF H2S F- Br+ N2 HCN NC-arrow_forwardAre these two compounds identical?arrow_forward
- Rank the following three bonds from most to least polar. P—Cl P—F P—Brarrow_forwardEach compound contains both ions and covalent bonds. Draw the Lewis structure for compound. Show with dashes which are covalent bonds and show with charges which are ions. Q.Sodium methoxide, CH3ONaarrow_forwardGive the systematic name of each covalent compound. Spelling counts. P2S5: C3N4:arrow_forward
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





