
Concept explainers
(a)
Interpretation:
Each carbon in the following molecule should be labeled as tetrahedral, trigonal planar or linear:
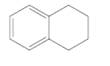
Concept Introduction:
Valence shell electron pair repulsion (VSEPR) theory is used to determine the shape. VSEPR is based on the fact that electron pairs repel each other; thus, when groups (a group is either an atom or a lone pair of electrons) are as far away from each other as possible then they are in most stable arrangement.
(b)
Interpretation:
Each carbon in the following molecule should be labeled as tetrahedral, trigonal planar or linear:
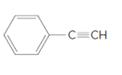
Concept Introduction:
Valence shell electron pair repulsion (VSEPR) theory is used to determine the shape. VSEPR is based on the fact that electron pairs repel each other; thus, when groups (a group is either an atom or a lone pair of electrons) are as far away from each other as possible then they are in most stable arrangement.
(c)
Interpretation:
Each carbon in the following molecule should be labeled as tetrahedral, trigonal planar or linear:
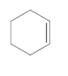
Concept Introduction:
Valence shell electron pair repulsion (VSEPR) theory is used to determine the shape. VSEPR is based on the fact that electron pairs repel each other; thus, when groups (a group is either an atom or a lone pair of electrons) are as far away from each other as possible then they are in most stable arrangement.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
- Why do alkanes, alkenes, and alkynes have a relatively low boiling point? O They have no large intermolecular forces holding them together. O They have no diple moments or polarity. O They have a lot of electron density in a very small amout of space.arrow_forward1. Predict the structure of ethanol once it oxidizes in the human body. Draw the condensed structure. 2. A student found a bottle in the lab filled with a colorless solution. The label on the bottle was fading. The students were able to correctly identify, but knewit was aa phenol from the readable part of the label. The student decided to run a ferric chloride test. The test resulted in a greed color. What is the identity of the liquid? 3. Predict below alcohols their skeletal structure; if soluble in water; does it contain a phenol group, is it primary, secondary, or tertiary alcohol; can it be oxidized by chromate; if yes what is the oxidation product and draw the skeletal structure; will the alcohol form a colorful ccomplex with Fe3+? Thymol ( found in oil of thyme, antiseptic properties) Menthol ( found in mint, gives a cooling sensation) Cinnamyl aalcohol (used in perfumes ans deodorants) Cety alcohol (ingredient in shampoo, creams, and lotions)arrow_forwardConvert each condensed formula to a Lewis structure. CH3(CH2)4CH(CH3)2 (CH3)3CCH(OH)CH2CH3 (CH3)2CHCHO (HOCH2)2CH(CH2)3C(CH3)2CH2CH3arrow_forward
- the bonds C-C, C-H, and C-O, in a molecule of acetone are polar covalent or nopolar covalent?arrow_forwardProvide the correct IUPAC name for the skeletal (line-bond) structure shown here. * 2,2,3,3-3,3- 2,3- 2,2,2,2- 3,3,3,3- tri di penta tetra eth hex meth 2,2-arrow_forwardWhat is the difference between the hybridization of carbon atoms' valence orbitals in saturated and unsaturated hydrocarbons?arrow_forward
- How many electron pairs are shared when a triple bond exists between two carbon atoms? What must he the geometric arrangement around the carbon atoms in a triple bond? Draw the Lewis structure of a simple molecule that contains a triple bond.arrow_forwardDraw a bond line structure for the following compouds. a. 1,1,2,2-Tetramethylcyclopropanearrow_forwardConvert each molecule into a skeletal structure. a. (CH3)½CHCH,CH2CH(CH3)2 c. CH3(CH2)½C(CH3)½CH(CH3)CH(CH3)CH(Br)CH3 нн HT TH ċ-C H CH3 c-c CH2 b. CH;CH(CI)CH(OH)CH3 d. CH3-C H limonene (oil of lemon)arrow_forward
- Which compound has the longest carbon-carbon bond length? All bond lengths are the same. HCCH CH3CH3 CH₂CH2arrow_forwardWhy can only up to 4 covalent bonds be formed with one carbon atom?arrow_forwardConvert each molecule into a skeletal structure. a. (CH3)2CHCH,CH;CH(CH3)2 c. CH3(CH2),C(CH3)½CH(CH3)CH(CH3)CH(Br)CH3 нн HT TH C-C H CH3 d. CH3-C C-C C-C b. CH3CH(CI)CH(OH)CH3 CH2 H limonene (oil of lemon)arrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





