
Concept explainers
(a)
Interpretation:
The
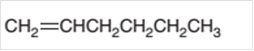
Concept Introduction:
A
In a chemical reaction, the substance which is involved in conversion is said to be reactant whereas the newly formed substance is known as a product. Both reactants and products must be separated by an arrow.
Hydrogenation reaction is an addition reaction in which the hydrogen atoms are bonded on un-statured carbon atoms of alkene to form alkane. Certain metal catalyst Ni and Pd are used for this reaction.
(b)
Interpretation:
The alkane formed on treating following alkene with H2 with Pd catalyst should be determined:
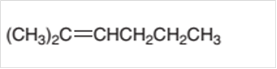
Concept Introduction:
A chemical reaction is the symbolic representation of the conversion of substances to new substances.
In a chemical reaction, the substance which is involved in conversion is said to be reactant whereas the newly formed substance is known as a product. Both reactants and products must be separated by an arrow.
Hydrogenation reaction is an addition reaction in which the hydrogen atoms are bonded on un-statured carbon atoms of alkene to form alkane. Certain metal catalyst Ni and Pd are used for this reaction.
(c)
Interpretation:
The alkane formed on treating following alkene with H2 with Pd catalyst should be determined:
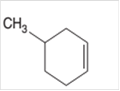
Concept Introduction:
A chemical reaction is the symbolic representation of the conversion of substances to new substances.
In a chemical reaction, the substance which is involved in conversion is said to be reactant whereas the newly formed substance is known as a product. Both reactants and products must be separated by an arrow.
Hydrogenation reaction is an addition reaction in which the hydrogen atoms are bonded on un-statured carbon atoms of alkene to form alkane. Certain metal catalyst Ni and Pd are used for this reaction.
(d)
Interpretation:
The alkane formed on treating following alkene with H2 with Pd catalyst should be determined:
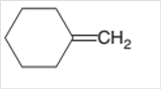
Concept Introduction:
A chemical reaction is the symbolic representation of the conversion of substances to new substances.
In a chemical reaction, the substance which is involved in conversion is said to be reactant whereas the newly formed substance is known as a product. Both reactants and products must be separated by an arrow.
Hydrogenation reaction is an addition reaction in which the hydrogen atoms are bonded on un-statured carbon atoms of alkene to form alkane. Certain metal catalyst Ni and Pd are used for this reaction.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
- What is the IUPAC name of the following compound? CH-CH, CH,CH,CHCH,CHCH,CH, ČHCH,CH, 3-ethylpropyl-1-heptene O ethyl-3-vinyloctene O 3,5-diethyl-1-octene O4,6-diethyl-1-octenearrow_forwardDraw the structures of organic molecules which fits each of these descriptions: An alkane An alkene An alcohol A carboxylic acid An aldehyde Draw the structures of each of these compounds: 2-bromopentane Trans-2-butene 1-bromo-3-nitrobenzene 4-chloro-pentane-2-ol 4-fluoro-1-methylcyclohexanearrow_forwardPart A - Naming Alkanes What is the proper name of this compound? ÇH3 CH,-CH,-C-CH3 CH2 ČH3 O 1,2-dimethyl-neopentane O 3,3-dimethylpentane O 2-ethyl-2-methylbutane O 3-ethyl-3-methylbutane O isoheptanearrow_forward
- Which alkane most readily undergoes thermal decomposition? Note that C-H bonds are usually stronger than C-C bonds. O ethane O dimethylpropane O propane O methylpropanearrow_forward3 Give the systematic name of the given alkene based on its semi-structural formula. CH, CH, CH3 CH, - C = CH — с %3 CH - CH, CH - - CH2 CH, Systematic name:arrow_forwardSelect the IUPAC name for the following structure: CH, CH-CH, ҪН,СH, CH,CH, CH—СH—СH—CH,—СH—СН, -CH—СH—CH, -CH-CH, CH—С—СH, CH, 3. O 4 isopropyl-2,2,3,6-tetramethyloctane O 2-ethyl-4-isopropyl-5,6,6-trimethylheptane O 4-isopropyl-2,2,3,6-tetramethyldecane . O 2-ethyl-4-isopropyl-5,6,6-trimethyloctane O 2-ethyl-4-isopropyl-5-isopropylhexanearrow_forward
- Three functional groups found in this compound are OH 0 CCH3 OCH3 O aldehyde, ether, and carboxylic acid O alcohol, aldehyde, and ether O alcohol, ether, and ketone O cycloalkene, alcohol, and carboxylic acidarrow_forwardWhich one of the following compounds is an alkene? CH3-CH2-0-CH2–CH3 || CH 3-CH 2-C -H OH CH3 -CH-CH 3 CH3-CH-CH-CH2–CH3 O CH3-S-Harrow_forward33) Cis-trans isomerism occurs when a branched alkane has a halogen added to two adjacent carbon atoms. an alkene is hydrated according to Markovnikov's Rule. the carbons in an alkene double bond have different substituent groups. the carbons in an alkene double bond each have the same substituent groups. hydrogen is added to both of the carbon atoms in a double bond.arrow_forward
- What is the molecular formula of a cyclic alkane with 6 carbon atoms? C6H14 C6H10 C6H16 O C6H12 соно Oarrow_forwardIndicate whether each statement is true or false. Alkanes do not have any carbon-carbon multiple bonds. Cyclobutane contains a four-membered ring. Alkenes contain carbon-carbon triple bonds. Alkynes contain carbon-carbon double bonds. Pentene is a saturated hydrocarbon. 1-pentene is an unsaturated hydrocarbon. Cyclohexane is an aromatic hydrocarbon. The methyl group contains one less hydrogen atom than methane.arrow_forwardDraw the condensed structural formula or skeletal formula, if yclic, for the alkene that is the major product from each of the following dehydration reactions: H+ а. CHз— CH—— СH — СH —ОН - Heat ОН ОН b. H+ с. H+ Heat Heat ОН d. CH3— CH — CH,— СH— СH, Нeat Draw the condensed structural formula for the ether produced y each of the following reactions: H+ а. 2СH3 — ОН Heat H+ b. 2CH3 — СH>— CH-— ОН Heatarrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co



