
Interpretation:
The carbon-carbon double bond should be labeled as cis or trans and IUPAC name of the alkene should be identified.
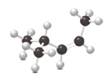
Concept Introduction:
Cis-trans isomers are fall under geometric isomerism.
Cis isomers- two different groups are on the same side of the double bond.
Trans isomers- two different groups are on opposite sides of the double bond.
In alkene nomenclature, longest C chain will be considered as the parent/ main C chain. At the end of the
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
- What type of hybridized orbital is present on carbon atoms bonded by a double bond? How many of these hybrid orbitals are on each carbon atom?arrow_forwardWhat type of hybridized orbital is present on carbon atoms bonded by a triple bond? How many of these hybrid orbitals are on each carbon atom?arrow_forwardProvide the IUPAC name for the given alkane. H₂C CH, Z CH₂CH₂ CH₂ CHarrow_forward
- An ether has the following structural formula: CH3-CH2-O-CH2-CH2-CH3. The IUPAC name isarrow_forward(1) Give the IUPAC name or common name of the isomer of 1-hexanol that has the highest vapor pressure (2) Give the IUPAC name of the alcohol that will produce 4-methylpentanal upon reaction with pyridinium chlorochromate.arrow_forwardGive the IUPAC name of each alkane below.arrow_forward
- Give the structural formula (condensed or skeletal) of the given IUPAC names of saturated hydrocarbons. 1) 2-methyl-3-oxohept-4-enoic acid 2) 4-chloro-2-oxohexanoyl chloride 3) 4-nitro-4-propoxybut-1-ene 4) 1-hydroxyhex-5-yn-2-one 5) 1-methylpropyl 3-amino-3-(1-methylpropoxy)propanoate 6) 1-ethoxy-3-mercaptohexane-2-thionearrow_forwardA saturated acyclic hydrocarbon has the molecular formula of CxH₂x2. A hydrocarbon can be classified as unsaturated if it has fewer hydrogens than a saturated formula would predict. Classify the following hydrocarbon skeletal structure as saturated or unsaturated. A) saturated B) unsaturatedarrow_forwardIndicate whether each statement is true or false: Alkenes contain carbon–carbon triple bonds.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning




