
Concept explainers
(a)
Interpretation:
An
Concept introduction:
E2 stands for bimolecular elimination. This reaction is a one-step concerted mechanism. In this step, the C-X and C-H bond breaks to form a double bond. The leaving group and the adjacent hydrogen atom must be anticoplanar in the precursor for the E2 step to be favored. To arrive back at the alkyl halide, one must add hydrogen and a halogen on the alkene carbons in an anti-fashion.
Since the alkyl halide and base influence the
Answer to Problem 13.35P
An alkyl halide that could have been used to synthesize the known alkene exclusively via an E2 reaction paying attention to stereochemistry is:
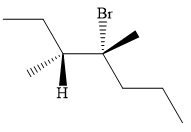
Explanation of Solution
The structure of the desired alkene is:
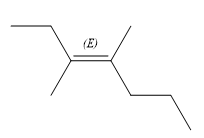
The given alkene has both the higher priority groups attached on the opposite side of the double bond. Hence, the stereochemistry is E. Alkenes can be prepared from corresponding alkyl halides via E2 reactions. This reaction is a one-step concerted mechanism. In this step, the C-X and C-H bond breaks to form a double bond. The leaving group and the adjacent hydrogen atom must be anticoplanar in the precursor for the E2 step to be favored. To arrive back at the alkyl halide, one must add hydrogen and a halogen on the alkene carbons in an anti-fashion. Thus, the original alkyl halide that could have been used to prepare the given alkene must be:

To arrive back at the alkyl halide, one must add hydrogen and a halogen on the alkene carbons in an anti-fashion.
(b)
Interpretation:
An alkyl halide, that can be used to synthesize the given alkene exclusively via an E2 reaction paying attention to stereochemistry, is to be provided.
Concept introduction:
The name E2 represents bimolecular elimination. This reaction is a one-step concerted mechanism. In this step, the C-X and C-H bond breaks to form a double bond. The leaving group and the adjacent hydrogen atom must be anticoplanar in the precursor for the E2 step to be favored. To arrive back at the alkyl halide, one must add hydrogen and a halogen on the alkene carbons in an anti-fashion.
Since the alkyl halide and base influence the rate of reaction, this is a bimolecular reaction. A strong base is used to form the most substituted alkene as the major product.
Answer to Problem 13.35P
An alkyl halide that could have been used to prepare the given alkene exclusively via an E2 reaction paying attention to stereochemistry is:
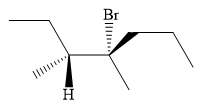
Explanation of Solution
The structure of the required alkene is:
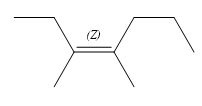
The given alkene has both the most priority groups attached on the same side of the double bond. Therefore, the stereochemistry about the double bond is Z. Alkenes can be prepared from corresponding alkyl halides via E2 reactions. This reaction is a one-step concerted mechanism. In this step, the C-X and C-H bond breaks to form a double bond. The leaving group and the adjacent hydrogen atom must be anticoplanar in the precursor for the E2 step to be favored. To arrive back at the alkyl halide, one must add hydrogen and a halogen on the alkene carbons in an anti-fashion. Thus, the original alkyl halide that could have been used to prepare the given alkene must be:

To arrive back at the alkyl halide, one must add hydrogen and a halogen on the alkene carbons in an anti-fashion.
(c)
Interpretation:
An alkyl halide, that can be used to synthesize the given alkene exclusively via an E2 reaction paying attention to stereochemistry, is to be provided.
Concept introduction:
The name E2 represents bimolecular elimination. This reaction is a one-step concerted mechanism. In this step, the C-X and C-H bond breaks to form a double bond. The leaving group and the adjacent hydrogen atom must be anticoplanar in the precursor for the E2 step to be favored. To arrive back at the alkyl halide, one must add hydrogen and a halogen on the alkene carbons in an anti-fashion.
Since the alkyl halide and base influence the rate of reaction, this is a bimolecular reaction. A strong base is used to form the most substituted alkene as the major product.
Answer to Problem 13.35P
An alkyl halide that could have been used to prepare the given alkene exclusively via an E2 reaction paying attention to stereochemistry is:
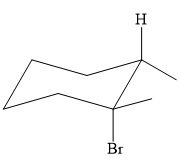
Explanation of Solution
The structure of the desired alkene is:
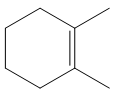
The alkene is a cycloalkene having two methyl groups attached to the double-bonded carbon atoms. As the carbon atoms in alkenes are

To arrive back at the alkyl halide, one must add hydrogen and a halogen on the alkene carbons in an anti-fashion.
(d)
Interpretation:
An alkyl halide, that can be used to synthesize the given alkene exclusively via an E2 reaction paying attention to stereochemistry, is to be provided.
Concept introduction:
The name E2 represents bimolecular elimination. This reaction is a one-step concerted mechanism. In this step, the C-X and C-H bond breaks to form a double bond. The leaving group and the adjacent hydrogen atom must be anticoplanar in the precursor for the E2 step to be favored. To arrive back at the alkyl halide, one must add hydrogen and a halogen on the alkene carbons in an anti-fashion.
Since the alkyl halide and base influence the rate of reaction, this is a bimolecular reaction. A strong base is used to form the most substituted alkene as the major product.
Answer to Problem 13.35P
An alkyl halide that could have been used to prepare the given alkene exclusively via an E2 reaction paying attention to stereochemistry is:
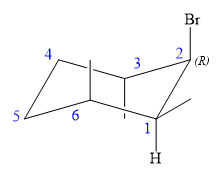
Explanation of Solution
The structure of the desired alkene is:
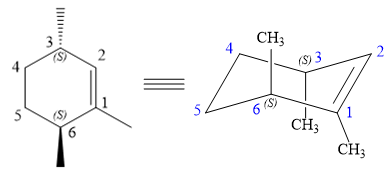
The alkene is cyclohexene having three methyl groups as substituents.
As the carbon atoms in alkenes are sp2 hybridized, all the atoms that are directly attached to the double-bonded carbon atoms must be in the plane. There are two chiral centers in the molecule at C3 and C6 carbon atoms. The stereochemistry for those two chiral centers will not change and will be retained as the reaction does not occur at those two chiral centers. Alkenes can be prepared from corresponding alkyl halides via E2 reactions.
This reaction is a one-step concerted mechanism. In this step, the C-X and C-H bond breaks to form a double bond. The leaving group and the adjacent hydrogen atom must be anticoplanar in the precursor for the E2 step to be favored. To arrive back at the alkyl halide, one must add hydrogen and a halogen on the alkene carbons in an anti-fashion. Thus, the original alkyl halide that could have been used to prepare the given alkene must be:
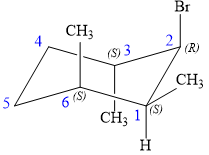
To arrive back at the alkyl halide, one must add hydrogen and a halogen on the alkene carbons in an anti-fashion.
(e)
Interpretation:
An alkyl halide, that can be used to synthesize the given alkene exclusively via an E2 reaction paying attention to stereochemistry, is to be provided.
Concept introduction:
The name E2 represents bimolecular elimination. This reaction is a one-step concerted mechanism. In this step, the C-X and C-H bond breaks to form a double bond. The leaving group and the adjacent hydrogen atom must be anticoplanar in the precursor for the E2 step to be favored. To arrive back at the alkyl halide, one must add hydrogen and a halogen on the alkene carbons in an anti-fashion.
Since the alkyl halide and base influence the rate of reaction, this is a bimolecular reaction. A strong base is used to form the most substituted alkene as the major product.
Answer to Problem 13.35P
An alkyl halide that could have been used to prepare the given alkene exclusively via an E2 reaction paying attention to stereochemistry is:
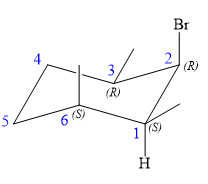
Explanation of Solution
The structure of the desired alkene is:
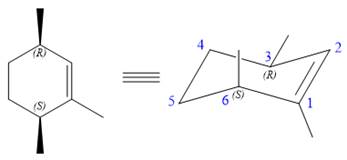
The alkene is cyclohexene having three methyl groups as substituents.
As the carbon atoms in alkenes are sp2 hybridized, all the atoms that are directly attached to the double-bonded carbon atoms must be in the plane. There are two chiral centers in the molecule at C3 and C6 carbon atoms. The stereochemistry for those two chiral centers will not change and will be retained as the reaction does not occur at those two chiral centers. Alkenes can be prepared from corresponding alkyl halides via E2 reactions.
This reaction involves a one-step mechanism (concerted) in which carbon-halogen bond and carbon-hydrogen bond breaks to form a double bond. The leaving group and the adjacent hydrogen atom must be anticoplanar in the precursor for the E2 step to be favored. To arrive back at the alkyl halide, one must add hydrogen and a halogen on the alkene carbons in an anti-fashion. Thus, the original alkyl halide that could have been used to synthesize the given alkene must be:
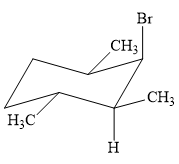
To arrive back at the alkyl halide, one must add hydrogen and a halogen on the alkene carbons in an anti-fashion.
Want to see more full solutions like this?
Chapter 13 Solutions
ORG.CHEM W/TEXT+SOLU.MANUAL
- (SYN) In the reaction shown here, the aromatic ring has just one chemically distinct, aromatic H, so a single electrophilic aromatic substitution will lead to just a single product. With this in mind, supply the missing reagentsneeded to carry out the transformation.arrow_forwardDraw the mechanism and the major organic product for each of the following reactions. Please show all work with arrowsarrow_forwardPropose a mechanism for the reaction shown here, which takes place under conditions that favor anarrow_forward
- Determine the major product of each reaction in Problem and draw the complete, detailed mechanism. Pay attention to stereochemistry where appropriate.arrow_forwardOCHEM help... What is the major product of the following reaction sequence? (See attached image)arrow_forwardDraw a mechanism to account for the reaction shown here, which scrambles the isotopic labelingarrow_forward
- ochem help please... What is the major product of the following reaction sequence? (see attached image)arrow_forwardTextbook problem: Suggest short series of reactions that would be expected to transform the material on the right into the desired product shown on left.arrow_forwardPlease draw detailed mechanism of this reaction. Picture are attached:arrow_forward
- Can you please help with the following organic chemistry reaction (see attached image) Provide the mechanism involved in the reaction and what the major product(s) would be. Thank youarrow_forwardIllustrate details mechanism of reactions for bromination of the following alkane. State each of the steps involves.arrow_forwardBy following the curved red arrows, draw the product(s) of each of the following reaction steps. Also indicate which species is the electrophile and which is the nucleophile.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
