
Concept explainers
Propose a structure consistent with each set of data.
a. a compound that contains a benzene ring and has a molecular ion at
b. a hydrocarbon that contains only
c. a compound that contains a carbonyl group and gives a molecular ion at
d. a compound that contains
(a)
Interpretation: The structure of a compound that contains a benzene ring and has a molecular ion at
Concept introduction: Molecular mass is the sum of the atomic weights of each constituent element multiplied by the number of atoms of that element.
Answer to Problem 13.37P
The structure of the compound that contains a benzene ring and has a molecular ion at
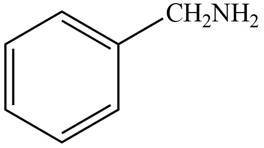
Explanation of Solution
The molecular ion peak of a compound is observed at
Possible hydrocarbons are calculated as,
• Divide
Thus, the possible molecular formula is
The molecular ion has odd mass. Thus, the compound may contain
Possible compounds with
• Substitute
Thus, the possible molecular formula is
The molecular mass of
Hence, the structure of a compound that contains a benzene ring and has a molecular ion at
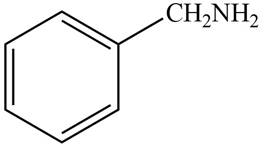
Figure 1
The structure of a compound
(b)
Interpretation: The structure of a hydrocarbon that contains only
Concept introduction: Molecular mass is the sum of the atomic weights of each constituent element multiplied by the number of atoms of that element.
Answer to Problem 13.37P
The structure of a hydrocarbon
Explanation of Solution
The molecular ion peak of a compound is observed at
Possible hydrocarbons are calculated as,
• Divide
• Replace one carbon by
Thus, the possible molecular formula is
The molecular mass of
Hence, the structure of a hydrocarbon that contains only
Figure 2
The structure of a hydrocarbon
(c)
Interpretation: The structure of a compound that contains a carbonyl group and gives a molecular ion at
Concept introduction: Molecular mass is the sum of the atomic weights of each constituent element multiplied by the number of atoms of that element.
Answer to Problem 13.37P
The structure of a compound
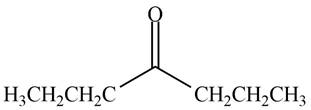
Explanation of Solution
The molecular ion peak of a compound is observed at
Possible hydrocarbons are calculated as,
• Divide
• Replace one carbon by
The compound that contain
Possible compounds with
• Substitute
Thus, the possible molecular formula is
Hence, the structure of a compound that contains a carbonyl group and gives a molecular ion at
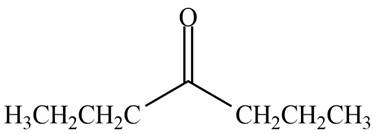
Figure 3
The structure of a compound
(d)
Interpretation: The structure of a compound that contains
Concept introduction: Molecular mass is the sum of the atomic weights of each constituent element multiplied by the number of atoms of that element.
Answer to Problem 13.37P
The structure of a compound
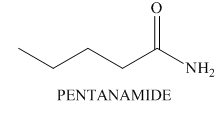
Explanation of Solution
The molecular ion peak of a compound is observed at
Possible hydrocarbons are calculated as,
• Divide
• Replace one carbon by
The given compound contains
Possible compounds with
• Substitute
• Substitute
Thus, the possible molecular formula is
Hence, the structure of a compound that contains

Figure 4
The structure of a compound
Want to see more full solutions like this?
Chapter 13 Solutions
ORGANIC CHEMISTRY-STUDY GUIDE PACKAGE
Additional Science Textbook Solutions
Introductory Chemistry (6th Edition)
Elementary Principles of Chemical Processes, Binder Ready Version
Chemistry
General, Organic, & Biological Chemistry
- Below are three MS spectra, Spectra 1, 2, and 3. Each of this mass spectrum corresponds to either Compound A (contains one Cl in the molecular formula), Compound B (one I in the molecular formula), and Compound C (one Br in the molecular formula). Identify which spectra corresponds to which compound. Provide explanations for your choices.arrow_forwardThe infrared spectrum of the compound with the mass spectrum shown below lacks any significant absorption above 3000 cm-1. There is a distinct peak near 1740 cm-1 and another strong peak near 1200 cm-1. molecular ion: M+ = 172arrow_forwardGiven the IR chart, identify what functional groups are present using the values below. Functional Group cm-1 O-H 3600-3200 N-H 3500-3200 Csp-H 3300 Csp2-H 3150-3000 Csp3-H 3000-2850 O=C-H 1740-1720 C=O 1800-1650 C=C (aromatic) 1650arrow_forward
- Propose a structure consistent with each set of spectral data assign each peak in the structure of your compoundarrow_forwardThe mass spectrum of 1-ethyl-1-methylcyclohexane shows many fragments, with two in very large abundance. Kne appears af m/z=111 and the other appears at m/z=97. Identify the structure of each of these fragments.arrow_forwardAn unknown compound (x) contains only carbon and hydrogen, has MW=112 and exhibits the spectral data below. In addition to the IR signal listed below, there are only peaks corresponding to C-H stretches (between 3300 and 2900) and several in the "fingerprint region". IR (cm-1) 2145 CNMR (ppm) 77.8, 70.1, 30.2 H NMR (ppm) 2.45 (singlet)arrow_forward
- Here are proton NMR data for 1-bromopropane: Ha : triplet (2H) 3.32ppm; Hb : multiplet (2H)1.81ppm; Hc : triplet (3H) 0.93ppm. (Relative integrations shown in parentheses.) a. Through how many bonds can a hydrogen split another hydrogen? b. According to this splitting rule, does Ha split Hc ? c. Is your answer in part a) consistent with the multiplicity listed for peak clusters a and c? d. How many hydrogenssplit Hb ? e. Upon very close inspection of the proton NMR spectrum of 1-bromopropane, you wouldfind that peak cluster b has at least six peaks. Is this consistent with your answer in part d)? f. Speculate as to why any peak cluster with more than four peaks is listed simply as a"multiplet."arrow_forwardLong-range coupling between protons more than two carbon atoms apart is sometimes observed when bonds intervene. An example is found in 1-methoxy-l-buten-3-yne. Not only does the acetylenic proton, Ηa, couple with the vinylic proton Hb, it also couples with the vinylic proton Hc, four carbon atoms away. The data are: Construct tree diagrams that account for the observed splitting patterns of Ηa, Hb, and Hc.arrow_forwardCompound A, a hydrocarbon with M+=96 in its mass spectrum, has the 13C spectral data given below. On reaction with BH3, followed by treatment with basic H2O2. A is converted into B, whose, 13C spectral data are also given below. Propose structures for A and B. Compound A Broadband-decoupled l3C NMR: 26.8, 28.7, 35.7, 106.9, 149.7 DEPT-90: no peaks DEPT-135: no positive peaks; negative peaks at 26.8, 28.7, 35.7, 106.9 Compound B Broadband-decoupled 13C NMR: 26.1, 26.9, 29.9, 40.5, 68.2 DEPT-90: 40.5 DEPT-135: positive peak at 40.5 ; negative peaks at 26.1, 26.9. 29.9, 68.2arrow_forward
- Following are 1H-NMR spectra for compounds G, H, and I, each with the molecular formula C5H12O. Each is a liquid at room temperature, is slightly soluble in water, and reacts with sodium metal with the evolution of a gas. (a) Propose structural formulas of compounds G, H, and I. (b) Explain why there are four lines between 0.86 and 0.90 for compound G. (c) Explain why the 2H multiplets at 1.5 and 3.5 for compound H are so complex.arrow_forwardPropose structures for compounds that fit the following mass-spectral data: (a) A hydrocarbon with M+=132 (b) A hydrocarbon with M+=166 (c) A hydrocarbon with M+=84arrow_forwardThe hormone cortisone contains C, H, and O, and shows a molecular ion at M+=360.1937 by high-resolution mass spectrometry. What is the molecular formula of cortisone? (The degree of unsaturation for cortisone is 8.)arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


