
Organic Chemistry Plus Masteringchemistry With Pearson Etext, Global Edition
9th Edition
ISBN: 9781292151229
Author: Wade, LeRoy G.
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 13, Problem 13.47SP
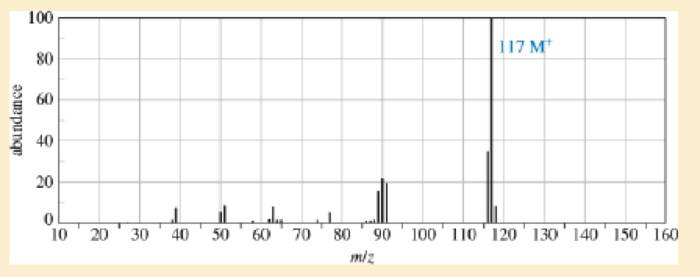
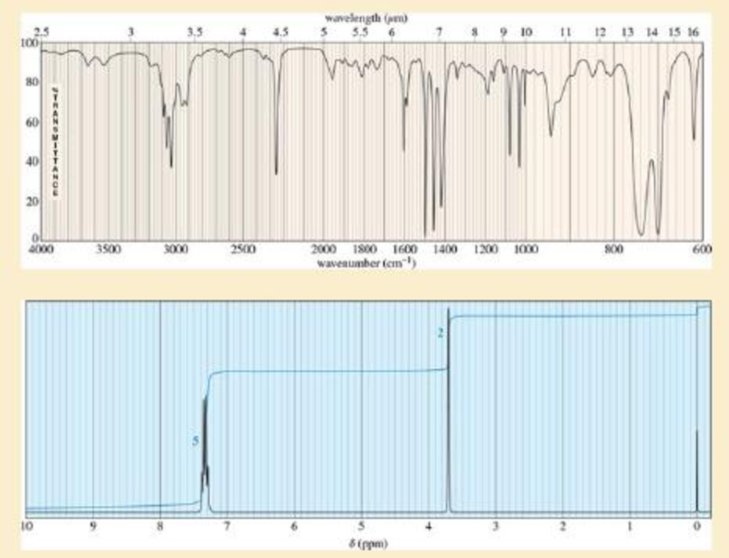
A compound was isolated as a minor constituent in an extract from garden cress. Its spectra are shown here.
- 1. Look at each spectrum individually, and list the structural characteristics you can determine from that spectrum.
- 2. Look at the set of spectra as a group, and propose a tentative structure.
- 3. Verify that your proposed structure accounts for the major features of each spectrum.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Organic chemistry: please explain this textbook question.
Predict the structure of the unknown compound using the provided information. All spectra provided for each unknown must be utilized to arrive at your answer. Please it should be clear how each spectra was used to help determine the unknown. Please annotate all spectra. Please provide the HDI and chemical formula and the final structure in bond line notation.
Deduce the stucture of compound E3 for which spectral data is provided. Make assignment for all the spectral peaks. Make these assignments in terms of structures or functionalities whenever possible.
Draw the proposed structure. If you can deduce two or more structures consistent with the data provided, indicate all of these structures and indicate your preference with any reasons you have for your choice.
4.) Determine and draw the structure corresponding to the spectral information provided below. As you make this determination, complete the table below. Explain what clues you used to determine the structure.
Chapter 13 Solutions
Organic Chemistry Plus Masteringchemistry With Pearson Etext, Global Edition
Ch. 13.5A - In a 300-MHz spectrometer, the protons in...Ch. 13.5B - Prob. 13.2PCh. 13.6 - Determine the number of different kinds of protons...Ch. 13.6 - Prob. 13.4PCh. 13.7 - Draw the integral trace expected for the NMR...Ch. 13.7 - Prob. 13.6PCh. 13.8C - Draw the NMR spectra you would expect for the...Ch. 13.8D - Draw the NMR spectra you expect for the following...Ch. 13.8D - a. Assign protons to the peaks in the NMR spectrum...Ch. 13.8D - Prob. 13.10P
Ch. 13.8D - Two spectra are shown. Propose a structure that...Ch. 13.9 - Prob. 13.12PCh. 13.9 - The spectrum of trans-hex-2-enoic acid follows. a....Ch. 13.9 - Prob. 13.14PCh. 13.9 - Prob. 13.15PCh. 13.10 - Prob. 13.16PCh. 13.10 - If the imaginary replacement of either of two...Ch. 13.10 - Predict the theoretical number of different NMR...Ch. 13.11B - Prob. 13.19PCh. 13.11B - Prob. 13.20PCh. 13.11B - Prob. 13.21PCh. 13.11B - Prob. 13.22PCh. 13.11B - Prob. 13.23PCh. 13.11B - Prob. 13.24PCh. 13.12E - Draw the expected broadband-decoupled 13 C N M R...Ch. 13.12E - a. Show which carbon atoms correspond with which...Ch. 13.12E - Repeat Problem13-25, sketching the...Ch. 13.12F - Prob. 13.28PCh. 13.13 - A bottle of allyl bromide was found to contain a...Ch. 13.13 - A laboratory student was converting cyclohexanol...Ch. 13.14 - Sets of spectra are given for two compounds. For...Ch. 13 - An unknown compound has the molecular formula C 9...Ch. 13 - Prob. 13.34SPCh. 13 - Predict the approximate chemical shifts of the...Ch. 13 - Prob. 13.36SPCh. 13 - Prob. 13.37SPCh. 13 - Prob. 13.38SPCh. 13 - Prob. 13.39SPCh. 13 - Prob. 13.40SPCh. 13 - For each compound shown below. 1. sketch the 13 C...Ch. 13 - Prob. 13.42SPCh. 13 - Prob. 13.43SPCh. 13 - Prob. 13.44SPCh. 13 - Prob. 13.45SPCh. 13 - Prob. 13.46SPCh. 13 - A compound was isolated as a minor constituent in...Ch. 13 - Prob. 13.48SPCh. 13 - The three isomers of dimethylbenzene are commonly...Ch. 13 - a. Draw all six isomers of formula C 4 H 8...Ch. 13 - Prob. 13.51SPCh. 13 - Hexamethylbenzene undergoes free-radical...Ch. 13 - Each of these four structures has molecular...Ch. 13 - Prob. 13.54SPCh. 13 - Phenyl Grignard reagent adds to 2-methylpropanal...Ch. 13 - Prob. 13.56SP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Propose a structure consistent with each set of spectral data assign each peak in the structure of your compoundarrow_forwardPLEASE EXPLAIN THIS TEXTBOOK QUESTION, SEE PHOTO: predict the structure of the unknown compound using the provided information. All spectra provided for each unknown must be utilized to arrive at your answer. Please it should be clear how each spectra was used to help determine the unknown. Please annotate all spectra. Please provide the HDI and chemical formula and the final structure in bond line notation.arrow_forwardCalculate the DoU for each of the following four spectral structures shown. Use spectral data to identify the four unknowns. You must provide structure of the unknown and provide information used to elicit the structure, ie. chemical shifts of protons, wavenumbers for functional groups and parent peak mass for molar mass of each compound. Povide an explanation for any work shown or drawn on your spectals. You must provide the name draw the structure of each which you propose in the space provided.arrow_forward
- The infrared and mass spectra of an unknown compound D are given below. Deduce the structure of this compound from these spectra.arrow_forwardPlease ensure all questions are answered it would be immensely appreciated!! Thanks The nmr spectra of three molecules are described below give the structure for the molecules and assign all peaks in the spectrum to protons in your molecule.arrow_forwardBased on the spectra provided, draw the structure. Label each unique carbon and hydrogen with the letters A, B, C… for use in assigning NMR peaks. fill in the data table assigning peaks in each spectrum. You should assign: • All 1H NMR peaks (do not worry about the peaks for CDCl3 at 7.26 ppm or the peak for TMS at 0 ppm) • Significant IR peaks above 1600 cm Example of what the table should include (imagine the structure of ethanol is drawn with the CH3 hydrogens labeled as A, the CH2 hydrogens labeled as B, and the OH hydrogen labeled as C) : hydrogen proton chemical shift integration splitting pattern couples to.. A 1.2 3 triplet B B 3.7 2 quartet A C 2.6 1 singlet -arrow_forward
- I'm about to have an organic chemistry test tomorrow and I don't fully understand the purpose of Mass Spectrometry. Can you please explain why it is used, is the molecule destroyed after (unlike IR spec), and any other useful details? I understand that IR spec is for identifying functional groups and together with NMR we can predict a likely compound.Where does mass spec come into play?Thank you!arrow_forwardI need to analyze the Ir spectrum and circle the frequency identify the groups and appearance strong board and weak on the spectrum graph I also need to draw a structure and match the peaks with its protons on the spectrumarrow_forwardCalculate the DoU for structures shown. Use spectral data to identify the four unknowns. You must provide structure of the unknown and provide information used to elicit the structure, ie. chemical shifts of protons, wavenumbers for functional groups and parent peak mass for molar mass of each compound. provide an explanation for any work shown or drawn on your spectals. You must provide the name draw the structure of each which you proposearrow_forward
- Given these 3 spectrums, draw the correct structure and explain step by step how you drew it.arrow_forwardOrganic Chemistry: IR spectroscopy A student obtains an IR spectrum with an intense, broad absorption at about 3350 cm-1. The student claims that the sample is an alcohol, but the professor says that the absorption is there because the sample is contaminated with water. What peak(s) could be looked for to show that the sample is indeed an alcohol? Which functional groups would make it difficult to use this strategy?arrow_forwardORGANIC CHEMISTRY: PLEASE EXPLAIN. AND SEE PHOTOpredict the structure of the unknown compound using the provided information. All spectra provided for each unknown must be utilized to arrive at your answer. Please it should be clear how each spectra was used to help determine the unknown. Please annotate all spectra. Please provide the HDI and chemical formula and the final structure in bond line notationarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY