
Concept explainers
(a)
Interpretation:
The given reaction equation to be predicted
Concept Introduction:
Addition reaction: A general reaction type in which a substance X-Y adds to the multiple bond of an unsaturated reactant to yield a saturated product that has only single bonds.
Hydrogenation (Addition of
(b)
Interpretation:
The given reaction equation to be predicted
Concept Introduction:
Addition reaction: A general reaction type in which a substance X-Y adds to the multiple bond of an unsaturated reactant to yield a saturated product that has only single bonds.
Halogenation (Addition of halogen molecule to alkenes): The addition of halogen molecules like
c)
Interpretation:
The given reaction equation to be predicted
Concept Introduction:
Addition reaction: A general reaction type in which a substance X-Y adds to the multiple bond of an unsaturated reactant to yield a saturated product that has only single bonds.
Hydrohalogenation (Addition of hydrogen halide to alkenes): The addition of hydrogen halide to a multiple bond to give an
Anti-Markovnikov’s rule: In the addition of
Markovnikov’s rule: In the addition of
(d)
Interpretation:
The given reaction equation to be predicted
Concept Introduction:
Addition reaction: A general reaction type in which a substance X-Y adds to the multiple bond of an unsaturated reactant to yield a saturated product that has only single bonds.
Hydration:
When alkene is undergoes hydration with water in the presence of sulfuric acid which yields the alcohol. In this reaction, the water molecule will behave like a hydrogen halide to the alkene which gives the addition product this reaction is known as a hydration reaction.
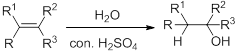
Alkene is reaction with water in the presence of sulfuric acid, first step is proton (
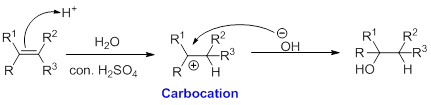
In hydration reaction, sulfuric acid is act as a proton donor, which is the driving force of the reaction. Hydration reaction will not go without acid (sulfuric acid).
Anti-Markovnikov’s rule: In the addition of
Markovnikov’s rule: In the addition of
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Fundamentals of General, Organic, and Biological Chemistry Plus Mastering Chemistry with Pearson eText -- Access Card Package (8th Edition)
- Is D-2-deoxygalactose the same chemical as D-2-deoxyglucose? Explain.arrow_forwardDraw a Fischer projection formula for the enantiomer of each of the following monosaccharides. (a to d)arrow_forwardWhat would be the characteristics of a transition-state analog for the chymotrypsin reaction?arrow_forward
