
Concept explainers
Can carbon dioxide be liquefied at room temperature
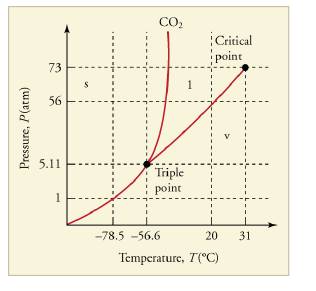
Figure 13.36 The phase diagram for carbon dioxide. The axes are nonlinear, and the graph is not to scale. Dry ice is solid carbon dioxide and has a sublimation temperature of -78.5°C .
Trending nowThis is a popular solution!

Chapter 13 Solutions
College Physics for AP Courses
Additional Science Textbook Solutions
College Physics: A Strategic Approach (3rd Edition)
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
Physics: Principles with Applications
Essential University Physics: Volume 2 (3rd Edition)
Conceptual Physics (12th Edition)
Sears And Zemansky's University Physics With Modern Physics
- Betong devises a customized temperature scale that assigns a temperature reading of 24 to the normal melting point of gold (1063°C) and a temperature reading of 299 to its boiling point (2808CC). What temperature on the Betong scale corresponds to absolute zero of temperature? Absolute zero in Celsius scale is -273°C. Round off answer to the nearest whole number. OB ANSWER:arrow_forwardA new linear temperature scale, degrees Zunzol (*Z), is based on the freezing point and boiling point of a newly discovered compound zunzol. The freezing point of zunzol, –117.3 °C, is defined as 0 °Z, and the boiling point of zunzol, 78.4 °C, is defined as 100 °Z. What is the freezing point of water in degrees Zunzol? freezing point: °Z What is the boiling point of water in degrees Zunzol? boiling point: °Zarrow_forwardProblem 1. Mercury (Hg) is used to measure temperatures due to its large volume expansivity. If the volume of Hg contained in a thermometer expanded from 0.7 cm³ at 10 °C to 0.707 cm³. What temperature should you read from the thermometer? The volume expansivity for mercury is: B= 181 x106/K. (10 points) Write your answer in the box provided below and show the work leading to that answer. T(°C) = 63.as enarrow_forward
- The vapor pressure of ethanol at 25°C is 0.07726 atm. Calculate the vapor pressure in kPa.Round answer to 4 significant digits.arrow_forwardYou just calibrated a constant volume gas thermometer. The pressure of the gas inside the thermometer is 254.0 kPa when the thermometer is in contact with boiling water. The boiling water is under regular atmospheric pressure. What is the temperature of the thermometer, when it reads a pressure of 494.0 kPa? Submit Answer Tries 0/12arrow_forwardA 10-kg chunk of ice at -30°C is mixed with 0.726 kg of water at 50°C. Find the temperature of the resulting mixture, assuming no heat gain or loss with the environment. Express your answer in degrees Celsius. (What can I use to solve this equation?) (I tried this and got 0, apparently that was the wrong answer.)arrow_forward
- A student constructs a simple constant volume gas thermometer and calibrates it using the boiling point of water, 100°C, and the freezing point of a specific brine solution, -21°C. The pressures measured at the calibration points are 1.366 atm and 0.9267 atm, respectively. From these data, what temperature will the student calculate, in degrees Celsius, for the value of absolute zero? T =arrow_forwardB. i. State the assumption of the kinetic theory of a gas that addresses molecular movement. ii. Given that: 3/2 kT = ½ m, where T = molecular temperature in kelvin and = molecular mean-square speed, sketch a general graph of T versus molecular root means square speed (c²>). jii. Given that for an ideal gas: PV = nRT, sketch a general graph of P versus T.arrow_forwardSuppose a walrus transfers energy by conduction through its blubber at the rate of 125 W when immersed in -1.25°C water. The walrus’s internal core temperature is 37.0°C, and it has a surface area of 1.85 m2. Chart attached. What is the average thickness of its blubber, in centimeters, which has the conductivity of fatty tissue?arrow_forward
- The normal freezing point of a certain liquid X is - 5.90 °C, but when 43. g of zinc chloride (ZnCl,) are dissolved in 400. g of X the solution freezes at -7.2 °C instead. Use this information to calculate the molal freezing point depression constant K, of X. Be sure your answer is rounded to the correct number of significiant digits. °C·kg K, = 0 molarrow_forwardQuestion 1. The table below shows a collection of data for a thermocouple. Temperature is in degrees Celsius, and the Voltage is in millivolts. (a) Plot the graph using rectangular graph paper with Temperature as the independent variable and determine the basic functional relationship between Temperature and Voltage. Alternatively, you can use Microsoft Excel to plot the data, but attach the graph in your answer page. (b) Determine specific the equation, using the method of selected points that best fits the data. Show how you determined the equation. If you need help, review Lecture-5 (c) What is the thermocouple voltage when the temperature is 1225 degree Celsius? Temperature (degree Celsius) Voltage (millivolts) 50 100 150 200 300 400 500 600 700 800 900 1000 2.5 6.7 8.8 11.2 17 22.5 26 32.5 37.7 41 48 55.2arrow_forwardHAND WRITTEN Plz..I'll upvote For a mixture of 40% nitrogen, 35% oxygen, and 25% carbon dioxide on a molar basis, what is the mixture constant a in the equation P=R ? Express your answer in kPa(m³/kmol)2K1/2 (+4)7arrow_forward
 College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON
University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press
Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley
Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON
College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON





