
Essential Organic Chemistry, Global Edition
3rd Edition
ISBN: 9781292089034
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 13, Problem 29P
(a)
Interpretation Introduction
Interpretation:
Aldehyde or ketone that is obtained when the given compound is heated in basic aqueous solution to be identified.
Concept introduction:
Aldol reaction is an addition reaction of
Mechanism for the aldol addition:
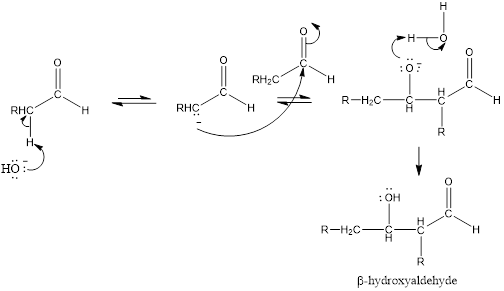
(b)
Interpretation Introduction
Interpretation:
Aldehyde or ketone that is obtained when the given compound is heated in basic aqueous solution to be identified.
Concept introduction:
Aldol reaction is an addition reaction of aldehydes and ketones.
Mechanism for the aldol addition:
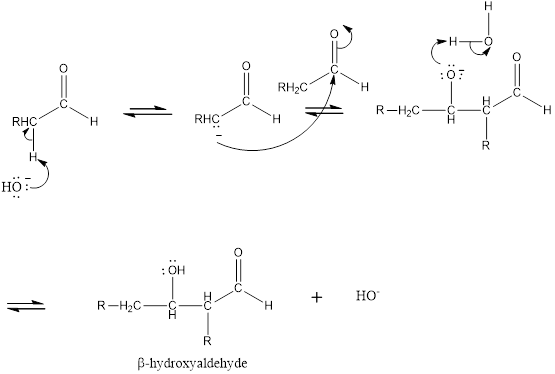
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What aldehyde or ketone would be obtained when each of the following compounds is heated in a basic aqueous solution? a. 2-ethyl-3-hydroxyhexanal c. 2,4-dicyclohexyl-3-hydroxybutanal b. 4-hydroxy-4-methyl-2-pentanone d. 5-ethyl-5-hydroxy-4-methyl-3-heptanone
4-Hydroxy- and 5-hydroxyaldehydes exist primarily as cyclic hemiacetals. Draw the structure of the cyclic hemiacetal formed by each of the following: a. 4-hydroxybutanal b. 4-hydroxypentanal c. 5-hydroxypentanal d. 4-hydroxyheptanal
Give the appropriate/simplest IUPAC name of the compound (see the attached picture)
A.) 5-tert-butyl-2-methylcyclopentanone
B.) 2-tert-butyl-5-methylcyclopentanone
C.) 2-tertbutyl-5-methylcyclopentanone
D.) 2-methyl-5-tertbutylcyclopentanone
E.) 5-methyl-2-tertbutylcyclopentanone
Chapter 13 Solutions
Essential Organic Chemistry, Global Edition
Ch. 13.1 - Identify the most acidic hydrogen in each...Ch. 13.1 - Prob. 2PCh. 13.1 - Prob. 3PCh. 13.1 - Prob. 4PCh. 13.1 - Explain why HO cannot remove a proton from the...Ch. 13.2 - Prob. 6PCh. 13.2 - Prob. 7PCh. 13.3 - Prob. 8PCh. 13.3 - Prob. 9PCh. 13.3 - Prob. 10P
Ch. 13.4 - Prob. 11PCh. 13.5 - Prob. 12PCh. 13.5 - Prob. 13PCh. 13.6 - Prob. 14PCh. 13.7 - Prob. 16PCh. 13.8 - Prob. 17PCh. 13.8 - Prob. 18PCh. 13.8 - Prob. 19PCh. 13.9 - Prob. 20PCh. 13.10 - Propose a mechanism for the formation of...Ch. 13.10 - Prob. 22PCh. 13.10 - a. If the biosynthesis of palmitic acid were...Ch. 13 - Draw the enol tautomers for each of the following...Ch. 13 - Number the following compounds in order from...Ch. 13 - Prob. 26PCh. 13 - Explain why the pKa of a hydrogen bonded to the...Ch. 13 - Prob. 28PCh. 13 - Prob. 29PCh. 13 - Prob. 30PCh. 13 - Prob. 31PCh. 13 - Prob. 32PCh. 13 - Prob. 33PCh. 13 - Using cyclopentanone as the reactant, show the...Ch. 13 - Prob. 35PCh. 13 - Prob. 36PCh. 13 - Prob. 37PCh. 13 - Prob. 38PCh. 13 - Prob. 39PCh. 13 - Prob. 40PCh. 13 - Prob. 41PCh. 13 - Prob. 42PCh. 13 - Prob. 43PCh. 13 - Prob. 44PCh. 13 - Describe how the following compounds can be...Ch. 13 - Prob. 46PCh. 13 - Which would require a higher temperature:...Ch. 13 - Prob. 48PCh. 13 - Propose a mechanism for the following reaction:Ch. 13 - Show how the following compounds could be...Ch. 13 - Prob. 51PCh. 13 - Prob. 52P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What aldehyde or ketone would be obtained when the following compound is heated in a basic aqueous solution? 4-hydroxy-4-methyl-2-pentanonearrow_forwardWhich of the following pair is correct to identify the molecule? a.)acetophenone: methyl phenyl ketone b.)2-pentanone: methyl propyl ketone c.)propanone: acetone d.)diphenyl ketone: benzophenonearrow_forwardWhat two functional groups react to form the following? a. A hemiacetal b. An acetal c. A ketal d. A hemiketalarrow_forward
- The following molecule belongs to a class of compounds called enediols; each carbon of the double bond carries an OH group. Draw structural formulas for the -hydroxyketone and the -hydroxyaldehyde with which this enediol is in equilibrium.arrow_forwardLabel each of the following structures as a cyclic hemiacetal, hemiketal, acetal, ketal, or none of these: a. b. c.arrow_forwardLabel each of the following structures as a hemiacetal, hemiketal, acetal, ketal, or none of these: a. c. b.arrow_forward
- Which of the following molecules gives a condensation reaction with benzaldehyde? a) methylbenzaldehyde b) 2,2-dimethylpropanoic acid c) 3-methylbenzaldehyde d) 4-chlorobenzaldehyde e) 3-methylbutanoic acidarrow_forwardWrite a balanced equation for the hydrogenation of eachof the following:a. Propanal (a three-carbon aldehyde)b. Propanone (a three-carbon ketone)c. 2,3-Dimethylheptanal (an aldehyde with a seven-carbon parent chain)d. 3-Methyl-4-heptanone (a ketone with a seven-carbonparent chain)arrow_forwardWhich compound will give a positive Tollen’s Test?arrow_forward
- Draw the structure for each of the following: a. isobutyraldehyde d. 3-methylcyclohexanone g. g-bromocaproaldehyde b. 4-hexenal e. 2,4-pentanedione h. 2-ethylcyclopentanecarbaldehyde c. diisopentyl ketone f. 4-bromo-3-heptanone i. 4-methyl-5-oxohexanalarrow_forwardIdentify the IUPAC name of the given compound. a) 2-methyl-3-amino-1-cyclopentanone b) 1-amino-2-methyl-3-cyclopentanone c) 3-amino-2-methyl-1-cyclopentanone d) 2-amino-1-methyl-2-cyclopentanonearrow_forwardWhich of the following compounds will form a yellow solid when dissolved in a basic, aqueous solution of I2?(a) butanoic acid; (b) pentan-2-one; (c) pentan-3-one; (d) cyclohexanone; (e) pentanalarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
How to Design a Total Synthesis; Author: Chemistry Unleashed;https://www.youtube.com/watch?v=9jRfAJJO7mM;License: Standard YouTube License, CC-BY