
Concept explainers
Interpretation:
Solid are the chemical substance which are characterised by definite shape and volume rigidity, high density, low compressibility. The constituent particles (atoms molecules or ions are closely packed and held together by strong force.
Types of solid:
The solid are of two types crystalline solids and amorphous solids.
Unit cell:
The smallest geometrical portion of the crystal lattice which can be as repetitive unit to build up the whole crystal is called unit cell.
Types of unit cell:
(i) Simple or primitive unit: In which the particle are present at the corners only.
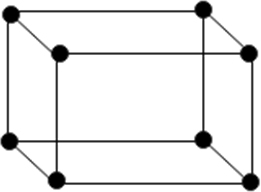
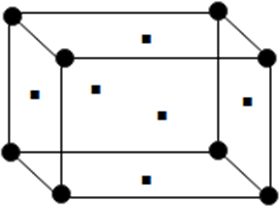
(ii) Face centred unit cell: In which the particle are present at the corners as well as at the centre of each of six faces.
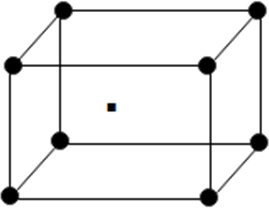
(iii) Body centred unit cell: In which the particles are present at the corners as well at the centre of unit.
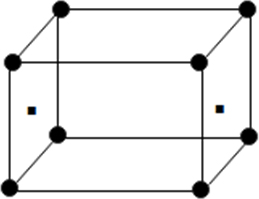
(iv) End centred unit cell: In which the particles are present at the corners and at the centre of two opposite faces.
Concept introduction:
No of particles and their contribution
| Unit cell | Corner | Face | Centre | Total |
| Body centred Unit cell |
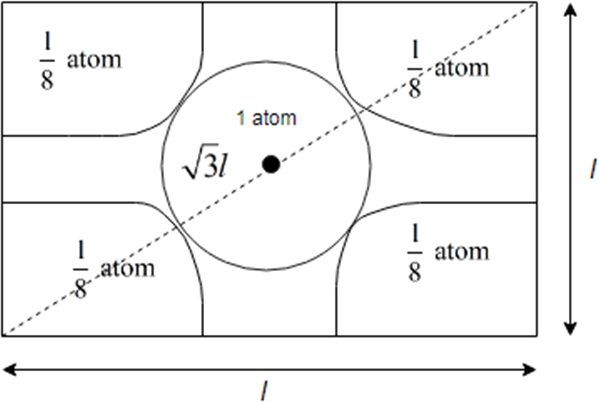
It is cross section view of body centered unit cell.
From above it becomes clear that body centred cubic unit cell atoms touch each other along the body diagonal
So length of body diagonal
To determine: the radius of the atom in pm in a body centered cubic unit cell that has volume of
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Study Guide For Chemistry: Structure And Properties
- Calcium oxide consists of a face-centered cubic array of O2 ions, with Ca2+ ions at the center of the unit cell and along the centers of all 12 edges. Calculate the number of each ion in the unit cell.arrow_forwardCrystalline polonium has a primitive cubic unit cell, lithium has a body-centered cubic unit cell, and calcium has a face-centered cubic unit cell. How many Po atoms belong to one unit cell? How many Li atoms belong to one unit cell? How many Ca atoms belong to one unit cell? Draw each unit cell. Indicate on your drawing what fraction of each atom lies within the unit cell.arrow_forwardSpinel is a mineral that contains 37.9% aluminum, 17.1% magnesium, and 45.0% oxygen, by mass, and has a density of 3.57 g/cm3.The edge of the cubic unit cell measures 809 pm. How many of each type of ion are present in the unit cell?arrow_forward
- The CsCl structure is a simple cubic array of chloride ions with a cesium ion at the center of each cubic array (see Exercise 69). Given that the density of cesium chloride is 3.97 g/cm3, and assuming that the chloride and cesium ions touch along the body diagonal of the cubic unit cell, calculate the distance between the centers of adjacent Cs+ and Cl ions in the solid. Compare this value with the expected distance based on the sizes of the ions. The ionic radius of Cs+ is 169 pm, and the ionic radius of Cl is 181 pm.arrow_forwardMn crystallizes in the same type of cubic unit cell as Cu. Assuming that the radius of Mn is 5.6% larger than the radius of Cu and the density of copper is 8.96 g/cm3, calculate the density of Mn.arrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





