
Interpretation:
The structure of an alcohol of given molecular formula C4H10O to be predicted using 13CNMR spectra.
Concept introduction:
The 13CNMR spectrum gives information on the different electronic environments of carbon. As like 1HNMR, the number of signals generated in 13CNMR are predicted by performing symmetry operations (rotation or reflection symmetry). Only chemical shift values are reported in the spectrum but not the multiplicity and integration values because the coupling between two neighboring 13C-13 C nuclei are weakly involved due to the low abundance of 13C isotopes of carbon atom.
To Identify:
The structure of an alcohol of given molecular formula C8H16O.
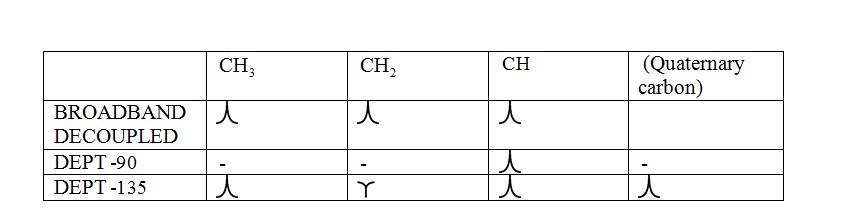
Broadband-decoupled 13CNMR spectrum:
The spectra provide information regarding the total number of carbon environments.
DEPT (Distortionless enhancement by polarization transfer):
a) DEPT-90: The spectrum exhibits signal only from CH group and no signals from CH3, CH2, CH and quaternary carbon (carbon with no protons).
b) DEPT-135: The spectrum exhibits CH3 groups and CH groups as positive signals (pointing up); CH2 groups appear as negative signals (pointing down) and quaternary carbon does not appear.
The signals appear in each type of spectrum:
Trending nowThis is a popular solution!

Chapter 13 Solutions
Organic Chemistry
- For each compound given below, determine whether the two labeled hydrogens are homotopic, enantiotopic, or diastereotopic.arrow_forwardWhat's the splitting pattern of the the bold carbon in C13-NMR? ( CH3)2CHCH2CH2OHarrow_forwardThe chemical shifts of the C-2 hydrogen in the spectra of pyrrole, pyridine, and pyrrolidine are 2.82 ppm, 6.42 ppm, and 8.50 ppm. Match each heterocycle with its chemical shift.arrow_forward
- What would be the chemical shift range of carboxylic acid protons? A. 4-6 ppm B. 6.5-8 ppm C. 2-5 ppm D. 10-12 ppmarrow_forwardAssign E or Z configuration to each of the following compounds:arrow_forwardHow many unique carbon atoms are in hexane (CH3CH2CH2CH2CH2CH3)? In other words, how many peaks would you expect in its 13C NMR spectrum? 1 2 3 6arrow_forward
- Draw the structure that fits each spectrum and assign each CHx group to appropriate peaks. (reference point at 0 ppm)arrow_forwardA sulfonium ion (R3S+) is a stereogenic center if three different alkyl groups are bonded to sulfur because sulfur is surrounded by four different groups, including its lone pair. In assigning an R or S) is a stereogenic center if three different alkyl groups are bonded to sulfur because sulfur is surrounded by four different groups, including its lone pair. In assigning an designation to sulfur, the lone pair is always assigned the lowest priority (4). SAM, S -adenosylmethionine, is a biologically active sulfonium ion that we will learn about in Section 7.16. Locate all the stereogenic centers in SAM, and assign an R,S designation to each center.arrow_forwardt please don't provide handwriting solution it's understandable that CH3 is ortho/para directing. but which of the two ortho positions would the Br stick to?arrow_forward
- Which group has the highest priority according to the Cahn, Ingold, Prelog rules? Select one: a. -CHO b. -OCOH c. -OCH3 d. - OCH2CH3 e. -COOCH3arrow_forwardHow many carbon atoms are present in a compound that gives an (M+) peak of 100% and a (M+1) peak of 13.2%?arrow_forwardAssign E or S configuration to each of the following compounds: a. b. c. d.arrow_forward
