
Concept explainers
(a)
Interpretation:
The following pair of

Concept Introduction:
Constitutional isomers are compounds which have the same molecular formula, but different connectivity of atoms. Stereoisomers are compounds which differ only in the three dimensional arrangement of atoms in space.
(b)
Interpretation:
The following pair of alkenes should be labeled as constitutional isomers or stereoisomers:

Concept Introduction:
Constitutional isomers are compounds which have the same molecular formula, but different connectivity of atoms. Stereoisomers are compounds which differ only in the three dimensional arrangement of atoms in space.
(c)
Interpretation:
The following pair of alkenes should be labeled as constitutional isomers or stereoisomers:
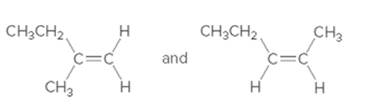
Concept Introduction:
Constitutional isomers are compounds which have the same molecular formula, but different connectivity of atoms. Stereoisomers are compounds which differ only in the three dimensional arrangement of atoms in space.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
CONNECT IA GENERAL ORGANIC&BIO CHEMISTRY
- Br2 ? How many moles of Br are required to completely halogenate the alkene? A. Three moles B. Four moles C. Two moles D. One mole What is the expected arrangement of the bromine atoms relative to each other among the carbon involved in pi bonding? A. trans-configuration B. anti-conformation C. cis-configuration D. syn-conformation What happens to bromine when it is adjacent to an alkene during a chemical reaction? A. Bromine becomes polarized. B. Bromine becomes acidic. C. Bromine becomes hybridized. D. Bromine becomes stable. The relative arrangement of bromine atoms in the product is primarily due to: A. Hydrogen bonding B. Electronegativity C. Atomic weight D. Repulsion What is your observation after the reaction? A. Bromine water decolorizes. B. A brown precipitate forms. C. The alkene becomes denser. D. A yellow flame is produced.arrow_forward[References] Br bromocyclopentane cyclopentyl acetate (85%) (15%) Propose a reaction mechanism for the formation of the minor product of this reaction by drawing the product of the following mechanistic step: HO CH3arrow_forwardHow many moles of Bra are required to completely halogenate the alkene?A. One moleB. Two molesC. Three molesD. Four moles What is the expected arrangement of the bromine atoms relative to each other amongthe carbon involved in pi bonding?A. anti-conformationB. syn-conformationC. trans-configurationD. cis-configuration What happens to bromine when it is adjacent to an alkene during a chemical reaction?A. Bromine becomes stable. (? kasi before brown siya/acidic tas naging colorless? Jk ewan)B. Bromine becomes polarized.C. Bromine becomes hybridized.D. Bromine becomes acidic. The relative arrangement of bromine atoms in the product is primarily due to:A. ElectronegativityB. RepulsionC. Hydrogen bondingD. Atomic weightWhat is your observation after the reaction?A. A yellow flame is produced.B. Bromine water decolorizes.C. The alkene becomes denser.D. A brown precipitate forms.arrow_forward
- Draw structural formulas for the major organic product of the reagents shown. CH3 • NO2 H2SO4 + HNO3 You do not have to consider stereochemistry. • Apply formal charges to any nitro groups. • If there is more than one major product possible, draw all of them. • • Separate multiple products using the + sign from the drop-down menu. ? √n [F ChemDoodleⓇarrow_forward3. Rank the following alkenes in decreasing order of stability, assigning number 1 to the most stable and number 4 to the least stable. Ph Ph Pharrow_forwardDraw what would happen to 2,5-dimethyl-2-hexene in an alkyl halides and alkane reaction. This is for a concept map. So what would the alkene 2,5-dimethyl-2-hexene be as an alkyl halides and how to you go from an alkene to an alkane with 2,5-dimethyl-2-hexene. No bond lines. I know this much for alkene to alkanearrow_forward
- 3. Which carbon would the Br of HBr add to in the following unsymmetrical alkenes? 1 Problem Set #7 Chemistry 227 а. b. С. d. H3C H3C е. H Н. H3C. TH. CH3 H H H the C=C in the ring do not react with H*arrow_forward4. Carbocations can undergo rearrangements to form more stable carbocations. A fourmembered ring can expand to a five-membered ring and a five-membered ring can expand to a six-membered ring, but a six-membered ring will not expand to a seven-membered ring. Provide an explanation for why the expansion of four- and five-membered rings is favorable, but the expansion of six-membered rings is not. O ** Carrow_forward1. Name the following alkenes, including stereochemistry.arrow_forward
- 1. Answer the following: a) What is the IUPAC name of the acyclic unbranched alkane that contains six carbons? b) How are structural isomers best defined? i. compounds with the same molecular formula whose atoms are bonded together in different arrangements ii. compounds with the same molecular formula whose atoms are bonded together in the same arrangement, but drawn differently iii. compounds with similar structural formulas, but different numbers of hydrogens iv. none of the above c) Which of the following statements is false concerning line structures of alkanes? i. Each carbon atom has 4 bonds, although they may not all be shown explicitly ii. The lines represent bonds between the carbon atoms. iii. Hydrogen atoms are not shown because they are not present in the molecule iv. none of the abovearrow_forwardDraw the product(s) of the following reactions. 1. BH3 / THF 2. H₂O₂/aqueous NaOH H3C-CEC-H • You do not have to consider stereochemistry. • Separate multiple products using the + sign from the drop-down menu. • You do not have to explicitly draw H atoms. • If no reaction occurs, draw the organic starting material.arrow_forwardWhich alkenes show cis,trans isomerism? For each alkene that does, draw the trans isomer. Q.) 2-Methyl-2-pentenearrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
